RHD Interventions
Primary Prevention. summarizes the key points of intervention in the natural history of ARF and RHD, covering primary and secondary prevention, surgical treatment, and primordial prevention, the latter referring to measures that reduce the incidence of streptococcal transmission in the general population. Research on primary prevention conducted in the 1950s among American military recruits demonstrated that penicillin treatment of streptococcal pharyngitis could reduce the risk of ARF by about 80 percent (Robertson, Volmink, and Mayosi 2005). Although most of the effectiveness data on primary prevention are older and of lower quality, penicillin is widely regarded as the mainstay of prevention and remains in all major clinical guidelines (Marijon and others 2012).
Major Categories of Interventions for the Prevention and Control of RHD.
Secondary Prevention. Early studies of individuals with a documented history of ARF demonstrated that regular secondary preventive therapy with penicillin—especially injectable benzathine penicillin—could reduce the risk of recurrent ARF and, by inference, RHD (Manyemba and Mayosi 2002). The rationale for secondary prevention is that it eliminates streptococcal colonization and thereby persistent subclinical inflammation and progressive valve damage (Majeed and others 1986). Sufficient evidence indicates that secondary prevention programs produce low rates of ARF recurrence in patients receiving continuous secondary prophylaxis. However, the quality of controlled studies is suboptimal, and it has been difficult to quantify the relationship between ARF recurrences averted and reductions in incident RHD (Manyemba and Mayosi 2002). Despite these evidence gaps, there is strong consensus globally that secondary prevention is effective and that further trials on its effectiveness would not be ethical.
Limitations of the Evidence for Prevention. From the policy standpoint, interpreting and applying the literature on primary and secondary prevention poses several challenges.
The studies are all of poor quality and are more than 20 years old; nearly all were conducted in HICs. These trials used older formulations of penicillin that are no longer in widespread use, limiting the usefulness of these data in contemporary economic models.
There is no evidence that primary or secondary prevention reduces RHD mortality, and no such trials are likely to be performed in children for ethical reasons.
No studies have been conducted for secondary prevention in adults with ARF and RHD, who constitute the majority of cases today.
An exclusive primary prevention strategy could miss a substantial proportion of cases because 50 percent to 75 percent of ARF cases may have no history of symptomatic pharyngitis.
Despite aggressive prevention efforts, many patients with established RHD require surgical intervention when valve dysfunction becomes severe and symptomatic (
Zühlke and others 2015).
Cardiac Surgery. For individuals with established RHD, surgical and percutaneous techniques are available to repair, replace, or palliate damaged valves. The mitral valve is most commonly affected by RHD and is the most frequent target of surgical and catheter-based interventions; the aortic and tricuspid valves are also susceptible. In general, patients with more than one valve involved have a poorer prognosis, even with adequate access to surgery (Marijon and others 2012).
For patients with isolated mitral stenosis (narrowed mitral valve) and favorable valve characteristics, catheter-based dilation (percutaneous balloon valvulotomy) has become the treatment standard—at least in settings with access to state-of-the-art equipment and interventional cardiologists. However, percutaneous procedures should be performed in centers with cardiothoracic surgical expertise in case of complications (). An alternative to percutaneous valvulotomy is closed mitral valvulotomy, which can be performed by a general or cardiothoracic surgeon in a center with fewer resources.
For many LMICs, however, the scale-up of open-heart surgical services may be the most important option for patients with advanced RHD. Given the prevalence of unfavorable mitral stenosis, mitral incompetence (which cannot currently be treated by catheter-based methods), and multivalvular disease, most patients with RHD are not eligible for minimally invasive techniques and eventually require surgical valve replacement. Valve replacement is palliative rather than curative; most patients require lifelong anticoagulation and are exposed to high complication rates (Marijon and others 2012).
Primordial Prevention. A final intervention for RHD, although theoretical at present, is a vaccine against group A streptococcus—primordial prevention. Vaccine research and development has been ongoing for years, with promising results in select populations from phase II clinical trials (Bisno and others 2005). Unfortunately, the global distribution of streptococcal serotypes is very different from those investigated in clinical trials (Steer and others 2009); an array of serotypes—more than could feasibly have been included in any previously developed multivalent vaccine—have been implicated in ARF. Efforts are underway to ensure the development of a vaccine that will be effective in LMICs (Dale and others 2013).
RHD Delivery Platforms
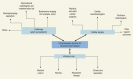
The potential delivery platforms for RHD-related interventions can be classified as follows:
Community-based efforts to educate children, parents, and educators about sore throat, ARF, and RHD
Provision of primary and secondary prophylaxis in outpatient settings, primarily in primary care settings
Third-level care at specialized or referral facilities that offer cardiology and cardiac surgery services.
Community-Based Primary and Secondary Prevention. Successful ARF and RHD programs have implemented a comprehensive approach that integrates community-based education and awareness with the scale-up of sore throat treatment to increase primary prevention and case finding of patients with ARF and RHD to build disease registers and increase secondary prevention. The WHO recommends a comprehensive approach to RHD control modeled after these types of programs (WHO 2004).
Unfortunately, as of 2012, ARF and RHD prevention had not been included in standard guidelines and protocols for child health, such as the Integrated Management of Childhood Illness program. This omission is partly because most child health programs focus on those under age five years, and streptococcal sore throat and ARF are uncommon in this group. Accordingly, although the RHD community has produced many resources for managing sore throat and developing secondary prevention programs (Wyber 2013), these resources have yet to be integrated with other child and adolescent health interventions. Partners in Health has developed an integrated model for noncommunicable diseases that includes RHD, factoring in such issues as registration, supply chain management, and adherence support at both first- and second-level hospitals (Partners in Health 2011). However, this model has not yet been applied in a broad range of settings.
Secondary Prevention Using Echocardiography. Following the publication of echocardiography screening studies (Marijon and others 2007), many research groups attempted to develop active case finding programs to increase secondary prevention using echocardiography in community and school settings.1 This approach was adopted by the Stop RHD A.S.A.P. Programme at the University of Cape Town (Robertson, Volmink, and Mayosi 2006) and by similar programs in the South Pacific (Lawrence and others 2013). Controversy remains about the long-term impact and cost-effectiveness of these programs because the natural history of cases detected by echocardiogram—and the effectiveness of secondary prophylaxis in this group—is unknown (Zühlke and Mayosi 2013).
Surgical Care Platforms. Although some countries have the capacity for specialized surgical and catheter-based interventions, at least in urban centers, the ratio of the population to the number of centers is grossly inequitable; only a handful of centers exist in all of Sub-Saharan Africa other than South Africa (Zühlke, Mirabel, and Marijon 2013). Three models of initiatives have helped ameliorate this situation:
Some well-selected cases are transferred for surgery on a philanthropic basis to Europe and the United States; a variant of this model is for visiting surgeons to set up temporary services in-country in conjunction with charitable organizations.
2Using South-South collaboration, patients are referred to high-volume regional or continental centers, such as in India or Sudan.
3 Unfortunately, many countries have national referral boards that finance out-of-country transfers on an extremely limited basis, and these referrals are likely to be somewhat biased against the rural poor who are less likely to receive a diagnosis or to benefit from advocacy efforts.
Lower-income countries start to build surgical platforms in their own countries (
Binagwaho and others 2013), although this model can be resource intensive and may detract from other health priorities.
ARF and RHD Public Policies for Prevention and Control
The WHO’s comprehensive set of guidelines on RF and RHD for LMICs (WHO 2004) recommended a package of several types of activities within an integrated RHD program (). The evidence for these public health initiatives largely came from Latin America and the Caribbean during the 1970s and 1980s, when ARF was essentially eradicated and the prevalence of severe RHD was dramatically reduced (Bach and others 1996; Nordet and others 2008). Although the decline in ARF and RHD in most regions has tracked closely with social and economic development, the role of primordial measures—policies dealing with risk factors such as overcrowding, sanitation and hygiene, and poor nutrition—is unclear, yet is likely to be significant (Gordis 1985).
Components of an Integrated Program on ARF and RHD Prevention and Control.
There have been recent efforts to develop policies for ARF and RHD prevention and control in Africa. A technical consultation initiated by the African Union in 2015 produced a set of seven key actions for ARF and RHD (Watkins, Zühlke, and others 2016). In addition to the elements recommended by the WHO (2004) report, this consultation stressed the need to ensure adequate supplies of high-quality penicillin, which has recently experienced poor availability globally. It also highlighted the many points of integration with reproductive and maternal health services and with other noncommunicable diseases. These recommendations have since been adopted in a resolution signed by all African heads of state, and implementation plans are currently being developed in collaboration with the WHO.