This is a work of the US government and distributed under the terms of the Public Domain
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
SUMMARY
Background:
Acesulfame potassium is an artificial sweetener widely used in beverages and foods. We tested if acesulfame potassium could cause cancer in two different strains of genetically modified mice.
Methods:
We fed groups of male and female Tg.AC mice and male and female p53 mice diets containing up to 3% acesulfame potassium for 9 months. Animals given feed with no sweetener added served as the control groups. Tissues from 15 sites were examined for every animal.
Results:
Exposure to acesulfame potassium had no effect on the survival of any of the animal groups. No increases in tumors were seen in males or females from either strain of mice.
Conclusions:
We conclude that acesulfame potassium did not cause cancer in the genetically modified mice used in these studies.
ABSTRACT
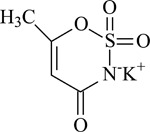
ACESULFAME POTASSIUM
CAS No. 55589-62-3
Chemical Formula: C4H4KNO4S Molecular Weight: 201.25
Synonyms: ASK; HOE-095K; 6-methyl-1,2,3-oxathiazine-4(3-H)-one-2,2-dioxide potassium salt
Trade names: Sunette, Sweet One
Acesulfame potassium is an artificial sweetener used throughout the world in food and beverages. Acesulfame potassium was nominated by The Center for Science in the Public Interest because of its widespread use. Male and female Tg.AC hemizygous and p53 haploinsufficient mice were exposed to acesulfame potassium (at least 99% pure) in feed for 9 months. Genetic toxicology studies were conducted in mouse peripheral blood erythrocytes.
9-Month Study in Tg.AC Hemizygous Mice:
Groups of 15 male and 15 female Tg.AC hemizygous mice were fed diets containing 0%, 0.3%, 1%, or 3% acesulfame potassium (equivalent to average daily doses of approximately 420, 1,400, or 4,500 mg acesulfame potassium/kg body weight to males and 520, 1,700, or 5,400 mg/kg to females) for 40 weeks. Exposure to acesulfame potassium had no effect on survival or mean body weights. Feed consumption by the exposed groups was similar to that by the control groups throughout the study. There were no neoplasms or nonneoplastic lesions that were attributed to exposure to acesulfame potassium.
9-Month Study in p53 Haploinsufficient Mice:
Groups of 15 male and 15 female p53 haploinsufficient mice were fed diets containing 0%, 0.3%, 1%, or 3% acesulfame potassium (equivalent to average daily doses of approximately 475, 1,500, or 4,700 mg/kg to males and 570, 1,800, or 5,700 mg/kg to females) for 40 weeks. Exposure to acesulfame potassium had no effect on survival or mean body weights. Feed consumption by the exposed groups was similar to that by the control groups throughout the study. There were no neoplasms or nonneoplastic lesions that were attributed to exposure to acesulfame potassium.
Genetic Toxicology:
Acesulfame potassium did not increase the frequency of micronucleated erythrocytes in peripheral blood of male or female Tg.AC hemizygous mice administered 0.3% to 3% in dosed feed. A similar study was conducted in p53 haploinsufficient mice, and a significant exposure concentration-related increase in the frequency of micronucleated erythrocytes was noted in males but not females.
Conclusions:
Under the conditions of this 9-month feed study, there was no evidence of carcinogenic activity* of acesulfame potassium in male or female p53 haploinsufficient mice exposed to 0.3%, 1%, or 3%.
Summary of the 9-Month Carcinogenesis and Genetic Toxicology Studies of Acesulfame Potassium
|
Male p53 Haploinsufficient Mice |
Female p53 Haploinsufficient Mice | |
|---|---|---|
| Concentrations in feed |
0%, 0.3%, 1%, or 3% (0, 3,000, 10,000, or 30,000 ppm) | 0%, 0.3%, 1%, or 3% |
| Body weights | Exposed groups similar to the control group | Exposed groups similar to the control group |
| Survival rates | 14/15, 15/15, 15/15, 14/15 | 14/15, 14/15, 14/15, 14/15 |
| Nonneoplastic effects | None | None |
| Neoplastic effects | None | None |
| Level of evidence of carcinogenic activity | No evidence | No evidence |
| Genetic toxicology | ||
| Micronucleated erythrocytes | ||
| Mouse peripheral blood in vivo: | Positive in p53 haploinsufficient males; negative in Tg.AC hemizygous males and females and p53 haploinsufficient females | |
- *
Explanation of Levels of Evidence of Carcinogenic Activity is on page 8. A summary of the Technical Report Review Subcommittee comments and public discussion on this technical report appears on page 9.
Contents
- FOREWORD
- CONTRIBUTORS
- EXPLANATION OF LEVELS OF EVIDENCE OF CARCINOGENIC ACTIVITY
- NATIONAL TOXICOLOGY PROGRAM BOARD OF SCIENTIFIC COUNSELORS TECHNICAL REPORTS REVIEW SUBCOMMITTEE
- SUMMARY OF TECHNICAL REPORTS REVIEW SUBCOMMITTEE COMMENTS
- INTRODUCTION
- MATERIALS AND METHODS
- RESULTS
- DISCUSSION AND CONCLUSIONS
- REFERENCES
- APPENDIX A. SUMMARY OF LESIONS IN MALE Tg.AC HEMIZYGOUS MICE IN THE 9-MONTH FEED STUDY OF ACESULFAME POTASSIUM
- APPENDIX B. SUMMARY OF LESIONS IN FEMALE Tg.AC HEMIZYGOUS MICE IN THE 9-MONTH FEED STUDY OF ACESULFAME POTASSIUM
- APPENDIX C. SUMMARY OF LESIONS IN MALE p53 HAPLOINSUFFICIENT MICE IN THE 9-MONTH FEED STUDY OF ACESULFAME POTASSIUM
- APPENDIX D. SUMMARY OF LESIONS IN FEMALE p53 HAPLOINSUFFICIENT MICE IN THE 9-MONTH FEED STUDY OF ACESULFAME POTASSIUM
- APPENDIX E. GENETIC TOXICOLOGY
- APPENDIX F. CHEMICAL CHARACTERIZATION AND DOSE FORMULATION STUDIES
- APPENDIX G. FEED AND COMPOUND CONSUMPTION IN THE 9-MONTH FEED STUDIES OF ACESULFAME POTASSIUM
About the Series
- NLM CatalogRelated NLM Catalog Entries
- NTP report on the toxicology studies of aspartame (CAS No. 22839-47-0) in genetically modified (FVB Tg.AC hemizygous) and B6.129-Cdkn2atm1Rdp (N2) deficient mice and carcinogenicity studies of aspartame in genetically modified [B6.129-Trp53tm1Brd (N5) haploinsufficient] mice (feed studies).[Natl Toxicol Program Genet Mod...]NTP report on the toxicology studies of aspartame (CAS No. 22839-47-0) in genetically modified (FVB Tg.AC hemizygous) and B6.129-Cdkn2atm1Rdp (N2) deficient mice and carcinogenicity studies of aspartame in genetically modified [B6.129-Trp53tm1Brd (N5) haploinsufficient] mice (feed studies).National Toxicology Program. Natl Toxicol Program Genet Modif Model Rep. 2005 Oct; (1):1-222.
- Toxicology studies of bromodichloromethane (CAS No. 75-27-4) in genetically modified (FVB Tg.AC Hemizygous) mice (dermal, drinking water, and gavage studies) and carcinogenicity studies of bromodichloromethane in genetically modified [B6.129-Trp53(tm1Brd) (N5) haploinsufficient] mice (drinking water and gavage studies).[Natl Toxicol Program Genet Mod...]Toxicology studies of bromodichloromethane (CAS No. 75-27-4) in genetically modified (FVB Tg.AC Hemizygous) mice (dermal, drinking water, and gavage studies) and carcinogenicity studies of bromodichloromethane in genetically modified [B6.129-Trp53(tm1Brd) (N5) haploinsufficient] mice (drinking water and gavage studies).National Toxicology Program. Natl Toxicol Program Genet Modif Model Rep. 2007 May; (5):1-227.
- NTP report on the toxicology studies of dichloroacetic acid (CAS No. 79-43-6) in genetically modified (FVB Tg.AC hemizygous) mice (dermal and drinking water studies) and carcinogenicity studies of dichloroacetic acid in genetically modified [B6.129-Trp53(tm1Brd) (N5) haploinsufficient] mice (drinking water studies).[Natl Toxicol Program Genet Mod...]NTP report on the toxicology studies of dichloroacetic acid (CAS No. 79-43-6) in genetically modified (FVB Tg.AC hemizygous) mice (dermal and drinking water studies) and carcinogenicity studies of dichloroacetic acid in genetically modified [B6.129-Trp53(tm1Brd) (N5) haploinsufficient] mice (drinking water studies).National Toxicology Program. Natl Toxicol Program Genet Modif Model Rep. 2007 Apr; (11):1-168.
- Review Toxicology and carcinogenesis studies of milk thistle extract (CAS No. 84604-20-6) in F344/N rats and B6C3F1 mice (Feed Studies).[Natl Toxicol Program Tech Rep ...]Review Toxicology and carcinogenesis studies of milk thistle extract (CAS No. 84604-20-6) in F344/N rats and B6C3F1 mice (Feed Studies).National Toxicology Program. Natl Toxicol Program Tech Rep Ser. 2011 May; (565):1-177.
- Review Toxicology and carcinogenesis studies of androstenedione (CAS No. 63-05-8) in F344/N rats and B6C3F1 mice (gavage studies).[Natl Toxicol Program Tech Rep ...]Review Toxicology and carcinogenesis studies of androstenedione (CAS No. 63-05-8) in F344/N rats and B6C3F1 mice (gavage studies).. Natl Toxicol Program Tech Rep Ser. 2010 Sep; (560):1, 7-31,33-171 passim.
- NTP Genetically Modified Model Report on the Toxicity Studies of Acesulfame Pota...NTP Genetically Modified Model Report on the Toxicity Studies of Acesulfame Potassium (CASRN 55589-62-3) in FVB/N-TgN(v-Ha-ras)Led (Tg.AC) Hemizygous Mice and Carcinogenicity Studies of Acesulfame Potassium in B6.129-Trp53tm1Brd (N5) Haploinsufficient Mice (Feed Studies)
Your browsing activity is empty.
Activity recording is turned off.
See more...
