Attribution Statement: LactMed is a registered trademark of the U.S. Department of Health and Human Services.
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Drugs and Lactation Database (LactMed®) [Internet]. Bethesda (MD): National Institute of Child Health and Human Development; 2006-.
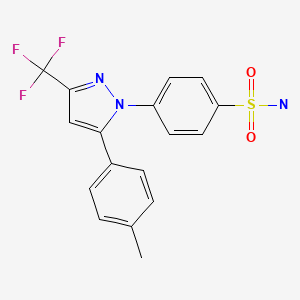
CASRN: 169590-42-5
Drug Levels and Effects
Summary of Use during Lactation
Because of the low levels of celecoxib in breastmilk, amounts ingested by the infant are small and would not be expected to cause any adverse effects in breastfed infants.[1] No special precautions are required.
Drug Levels
Maternal Levels. Milk celecoxib levels were measured in one mother taking 100 mg orally twice daily. At 4.75 hours after the fourth dose, milk levels were 101 and 133 mcg/L from her right and left breasts, respectively. A second peak level of about 30 mcg/L occurred in milk from the right breast about 35 hours after the last dose. The average half-life in the milk was 5.1 hours. The authors estimated that a fully breastfed infant would receive a maximum dose of 40 mcg/kg daily.[2]
Three mothers, averaging 12 months postpartum, who had been taking oral celecoxib 200 mg daily (average 3.27 mg/kg daily) for several weeks had milk celecoxib levels measured at several times during a 24-hour period. Peak milk levels averaged 139 mcg/L at 2 hours after the dose. By 12 hours after the dose, celecoxib was undetectable (<10 mcg/L) in milk. Two other mothers were given a single oral dose of celecoxib 200 mg. Breastmilk and maternal serum were collected periodically over an 8-hour period. Peak milk levels were 170 mcg/L at 4 hours after the dose in one woman and 269 mcg/L at 2 hours after the dose in the other.[3] Based on data from these 5 women, an exclusively breastfed infant would receive an estimated 9.8 mcg/kg daily or 0.3% of the maternal weight-adjusted dosage.
Six women averaging 11 months postpartum (range 6.5 to 15 months) were given a single 200 mg dose of celecoxib orally. Milk samples were collected for 48 hours after the dose. A median peak milk level of 200 mcg/L (range 70 to 330 mcg/L) occurred 2 to 4 hours after the dose. The authors calculated that a fully breastfed infant would receive a median daily dosage of 13 mcg/kg (range 11 to 210 mcg/kg) or 0.23% of the maternal weight-adjusted dosage.[4]
Thirteen nonlactating women took celecoxib 200 or 400 mg twice daily for 2 weeks followed by collection of nipple aspirate fluid. Two women taking 200 mg twice daily had celecoxib concentrations of 1.2 and 1.5 mg/L; 2 women taking 400 mg twice daily had celecoxib concentrations of 0.53 and 8.4 mg/L in the aspirate. Celecoxib was undetectable (lower limit of assay not stated) in the in nipple aspirate fluid of the other 9 women. Collection times with respect to the doses were not stated.[5]
Infant Levels. Two breastfed infants whose mothers had been taking celecoxib 200 mg orally twice daily for many weeks, had blood samples taken 4 hours after a maternal dose. The infants were 17 and 22 months of age and were being breastfed on demand every 3 to 4 hours during the day and once at night. Celecoxib was undetectable (<10 mcg/L) in the plasma of these infants.[3]
Effects in Breastfed Infants
No infant side effects were noted by the mothers of 2 infants aged 17 and 22 months who were breastfed during long-term maternal use of celecoxib 200 mg daily.[3]
Effects on Lactation and Breastmilk
Relevant published information was not found as of the revision date.
Alternate Drugs to Consider
Acetaminophen, Flurbiprofen, Ibuprofen, Indomethacin, Piroxicam
References
- 1.
- Götestam Skorpen C, Hoeltzenbein M, Tincani A, et al. The EULAR points to consider for use of antirheumatic drugs before pregnancy, and during pregnancy and lactation. Ann Rheum Dis. 2016;75:795–810. [PubMed: 26888948]
- 2.
- Knoppert DC, Stempak D, Baruchel S, et al. Celecoxib in human milk: A case report. Pharmacotherapy. 2003;23:97–100. [PubMed: 12523466]
- 3.
- Hale TW, McDonald R, Boger J. Transfer of celecoxib into human milk. J Hum Lact. 2004;20:397–403. [PubMed: 15479658]
- 4.
- Gardiner SJ, Doogue MP, Zhang M, et al. Quantification of infant exposure to celecoxib through breast milk. Br J Clin Pharmacol. 2006;61:101–4. [PMC free article: PMC1884977] [PubMed: 16390357]
- 5.
- Ruhlen RL, Chen YC, Rottinghaus GE, et al. RE: "Transfer of celecoxib into human milk. J Hum Lact. 2007;23:13–4. [PubMed: 17293545]
Substance Identification
Substance Name
Celecoxib
CAS Registry Number
169590-42-5
Disclaimer: Information presented in this database is not meant as a substitute for professional judgment. You should consult your healthcare provider for breastfeeding advice related to your particular situation. The U.S. government does not warrant or assume any liability or responsibility for the accuracy or completeness of the information on this Site.
- User and Medical Advice Disclaimer
- Drugs and Lactation Database (LactMed) - Record Format
- LactMed - Database Creation and Peer Review Process
- Fact Sheet. Drugs and Lactation Database (LactMed)
- Drugs and Lactation Database (LactMed) - Glossary
- LactMed Selected References
- Drugs and Lactation Database (LactMed) - About Dietary Supplements
- Breastfeeding Links
- PMCPubMed Central citations
- PubChem SubstanceRelated PubChem Substances
- PubMedLinks to PubMed
- Review Misoprostol.[Drugs and Lactation Database (...]Review Misoprostol.. Drugs and Lactation Database (LactMed®). 2006
- Review Bosentan.[Drugs and Lactation Database (...]Review Bosentan.. Drugs and Lactation Database (LactMed®). 2006
- Review Sildenafil.[Drugs and Lactation Database (...]Review Sildenafil.. Drugs and Lactation Database (LactMed®). 2006
- Review Nizatidine.[Drugs and Lactation Database (...]Review Nizatidine.. Drugs and Lactation Database (LactMed®). 2006
- Review Propranolol.[Drugs and Lactation Database (...]Review Propranolol.. Drugs and Lactation Database (LactMed®). 2006
- Celecoxib - Drugs and Lactation Database (LactMed®)Celecoxib - Drugs and Lactation Database (LactMed®)
Your browsing activity is empty.
Activity recording is turned off.
See more...
