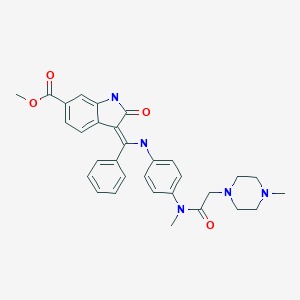
CASRN: 656247-17-5
Drug Levels and Effects
Summary of Use during Lactation
No information is available on the clinical use of nintedanib during breastfeeding. Because nintedanib is more than 97% bound to plasma proteins, the amount in milk is likely to be low. However, its half-life is about 9.7 hours and it might accumulate in the infant. The manufacturer recommends that breastfeeding be discontinued during nintedanib therapy.
Drug Levels
Maternal Levels. Relevant published information was not found as of the revision date.
Infant Levels. Relevant published information was not found as of the revision date.
Effects in Breastfed Infants
Relevant published information was not found as of the revision date.
Effects on Lactation and Breastmilk
Relevant published information was not found as of the revision date.
Substance Identification
Substance Name
Nintedanib
CAS Registry Number
656247-17-5
Drug Class
- Breast Feeding
- Lactation
- Antineoplastic Agents
- Enzyme Inhibitors
- Protein Kinase Inhibitors
- Signal Transduction Inhibitors
- Tyrosine Kinase Inhibitors
Disclaimer: Information presented in this database is not meant as a substitute for professional judgment. You should consult your healthcare provider for breastfeeding advice related to your particular situation. The U.S. government does not warrant or assume any liability or responsibility for the accuracy or completeness of the information on this Site.
Publication Details
Publication History
Last Revision: December 3, 2018.
Copyright
Attribution Statement: LactMed is a registered trademark of the U.S. Department of Health and Human Services.
Publisher
National Institute of Child Health and Human Development, Bethesda (MD)
NLM Citation
Drugs and Lactation Database (LactMed®) [Internet]. Bethesda (MD): National Institute of Child Health and Human Development; 2006-. Nintedanib. [Updated 2018 Dec 3].