Attribution Statement: LactMed is a registered trademark of the U.S. Department of Health and Human Services.
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Drugs and Lactation Database (LactMed®) [Internet]. Bethesda (MD): National Institute of Child Health and Human Development; 2006-.
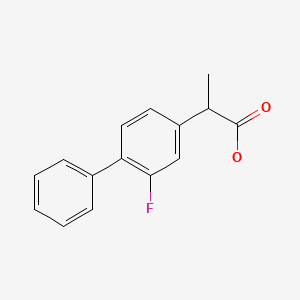
CASRN: 5104-49-4
Drug Levels and Effects
Summary of Use during Lactation
Because of the low levels of flurbiprofen in breastmilk and its short half-life it is unlikely to adversely affect the breastfed infant, especially if the infant is older than 2 months.
Maternal use of flurbiprofen eye drops would not be expected to cause any adverse effects in breastfed infants. To substantially diminish the amount of drug that reaches the breastmilk after using eye drops, place pressure over the tear duct by the corner of the eye for 1 minute or more, then remove the excess solution with an absorbent tissue.
Drug Levels
Maternal Levels. Ten women who were at least 1 month postpartum were given a single 100 mg tablet of flurbiprofen. Flurbiprofen was undetectable (<70 mcg/L) in the milk of 5 of the women. In the remaining 5 women, an average peak milk level of 92 mcg/L occurred at about 3 hours after the dose. Flurbiprofen was undetectable in any mother's milk by 24 hours after the dose.[1] Using data from this study, a fully breastfed infant would ingest an estimated maximum daily dosage of 2.2 mcg/kg after this maternal dosage or about 0.1% of the maternal weight-adjusted dosage.
Twelve women who were 3 to 5 days postpartum received flurbiprofen 50 mg orally four times a day for 9 doses over 3 days. Milk samples were collected at various unspecified times during this regimen. Flurbiprofen was undetectable (<50 mcg/L) in all but 3 milk samples. One woman had levels of 70 mcg/L 1 hour after her first dose and 60 mcg/L just before her second dose. Another mother had a milk flurbiprofen concentration of 80 mcg/L 2 hours after her last dose. The authors estimated that the maximum dose that a completely breastfed infant would ingest is less than 0.5% of the maternal weight-adjusted dosage.[2]
Infant Levels. Relevant published information was not found as of the revision date.
Effects in Breastfed Infants
A retrospective medical record review in Taiwan compared the full-term breastfed infants of women who received acetaminophen (n = 348) to those who received flurbiprofen (n = 132) for postpartum analgesia after a vaginal birth. There was no statistically significant difference in the percentage of infants with hyperbilirubinemia between those whose mothers received flurbiprofen (0.76%) and those whose mothers received acetaminophen(2.01%).[3]
A study of full-term, vaginally delivered breastfed neonates compared those whose mothers received acetaminophen (n = 348) to those whose mothers received flurbiprofen (n = 132) for postpartum pain. Seven (2%) newborns of acetaminophen users had hyperbilirubinemia and 1 (0.76%) newborn of the flurbiprofen users had hyperbilirubinemia. The differences was not statistically significant.[3]
Effects on Lactation and Breastmilk
Relevant published information was not found as of the revision date.
Alternate Drugs to Consider
(Systemic) Acetaminophen, Ibuprofen, Indomethacin, Naproxen, Piroxicam
References
- 1.
- Cox SR, Forbes KK. Excretion of flurbiprofen into breast milk. Pharmacotherapy. 1987;7:211–5. [PubMed: 3444752]
- 2.
- Smith IJ, Hinson JL, Johnson VA, et al. Flurbiprofen in post-partum women: Plasma and breast milk disposition. J Clin Pharmacol. 1989;29:174–84. [PubMed: 2715375]
- 3.
- Fu SC, Wang CY, Lin CK. Do postpartum nonsteroidal antiinflammatory drugs (NSAIDs) affect neonatal hyperbilirubinaemia? Taiwan J Obstet Gynecol. 2020;59:891–4. [PubMed: 33218407]
Substance Identification
Substance Name
Flurbiprofen
CAS Registry Number
5104-49-4
Disclaimer: Information presented in this database is not meant as a substitute for professional judgment. You should consult your healthcare provider for breastfeeding advice related to your particular situation. The U.S. government does not warrant or assume any liability or responsibility for the accuracy or completeness of the information on this Site.
- User and Medical Advice Disclaimer
- Drugs and Lactation Database (LactMed) - Record Format
- LactMed - Database Creation and Peer Review Process
- Fact Sheet. Drugs and Lactation Database (LactMed)
- Drugs and Lactation Database (LactMed) - Glossary
- LactMed Selected References
- Drugs and Lactation Database (LactMed) - About Dietary Supplements
- Breastfeeding Links
- PubChem SubstanceRelated PubChem Substances
- PubMedLinks to PubMed
- Flurbiprofen - Drugs and Lactation Database (LactMed®)Flurbiprofen - Drugs and Lactation Database (LactMed®)
- MIR506 microRNA 506 [Homo sapiens]MIR506 microRNA 506 [Homo sapiens]Gene ID:574511Gene
Your browsing activity is empty.
Activity recording is turned off.
See more...
