ABSTRACT
Growth hormone (GH) serves important roles in adult life, including maintenance of lean body mass and bone mass, promoting lipolysis, thereby limiting visceral adiposity, and regulating carbohydrate metabolism, cardiovascular system function, aerobic exercise capacity, and cognitive function. Younger adults with growth hormone deficiency (AGHD) exhibit abnormalities in body composition, physical and cognitive function, and quality of life which are reversed by GH replacement therapy. With advancing age GH production declines, paralleled by physical and functional alterations similar to those of AGHD; however, the degree to which the decrease in GH contributes to these age-related changes is unknown. Seemingly in opposition to the theory that the diminished GH secretion of older age is a net detriment are observations that animal models of congenital GH deficiency have remarkably increased life span and humans with congenital GH deficiency may have decreased rates of age-related diseases such as diabetes and cancer. Several short-term studies aiming to increase GH in older adults by a variety of interventions including exercise, administration of GH, or treatment with GH secretagogues have demonstrated consistent effects to improve body composition, yet inconsistent effects on physical and cognitive function. While side effects of GH administration in older adults include edema, arthralgias, and elevated blood glucose, data regarding the possible long-term effects on “hard end points” such as risk of fractures, cancer, cardiovascular disease, life expectancy, and mortality are lacking. For complete coverage of all related areas of Endocrinology, please visit our on-line FREE web-text, WWW.ENDOTEXT.ORG.
INTRODUCTION
The decline in growth hormone secretion observed with aging is associated with changes in body composition and physical and psychological function that are similar to those seen in younger adult patients with growth hormone deficiency. These changes include reductions in lean body mass and muscle strength and an increase in body fat, particularly in the visceral compartment. Memory and cognitive function gradually deteriorate with age. Deep (slow-wave) sleep also decreases markedly with age, together with a decrease in nighttime growth hormone secretion, and sleep disorders become a significant clinical problem in old age. Although these changes only show an association, and it is still unknown whether there is any causal link between them, they have led to speculation that replacing or stimulating growth hormone may reverse some of the detrimental features of the aging process (1).
AGE-RELATED CHANGES IN GROWTH HORMONE SECRETION
The trophic hormones which rise at puberty, including sex steroids and GH, have dramatic effects on body composition and strength. Their levels plateau in young adult life and then decline progressively, accompanied by a loss of muscle mass and aerobic capacity, and an increase in abdominal fat. These changes resemble some features of hypogonadism and adult GH deficiency (2).
After the third decade of life, there is a progressive decline of GH secretion by approximately 15% for every decade of adult life. Integrated measurements of daily GH secretion demonstrate that secretion peaks at puberty at about 150 µg/kg day, then decreases to approximately 25 µg/kg/day by age 55 (3). The reduction in GH secretion results from a marked reduction in GH pulse amplitude, with only very little change in pulse frequency (4). This process is characterized by lack of day-night GH rhythm resulting from loss of nocturnal sleep-related GH pulses (Figure 1) (4). Growth Hormone Binding Protein decreases from 60 years of age, theoretically increasing the amount of bioavailable growth hormone (5). This decrease is thought to parallel the decrease in growth hormone receptors with age. Though slow-wave sleep (SWS) decreases with age most studies administering GH or GHRH to seniors did not improve SWS. This finding suggests that the age-related decline in GH does not cause reduced SWS, although the reverse may be true.
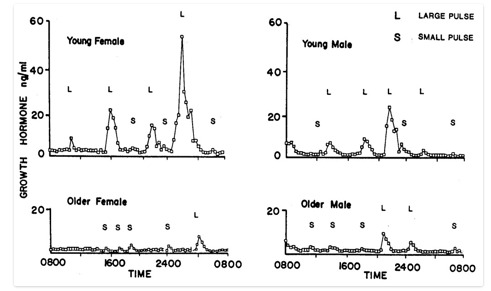
FIGURE 1.
Patterns of GH secretion in younger and older women and men. There is a marked age-related decline in GH secretion in both sexes and a loss of the nighttime enhancement of GH secretion seen during deep (slow-wave) sleep. This decrease is primarily due to a reduction in GH pulse amplitude, with little change in pulse frequency. L = large GH pulses, S = small GH pulses. From Ho et al. 1987 (4).
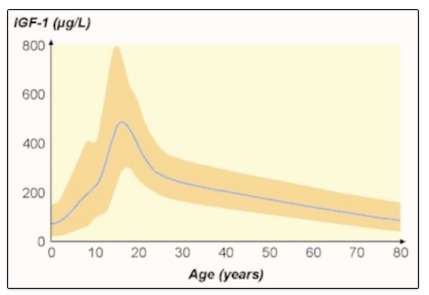
Circulating levels of IGF-I, the main mediator for the trophic effects of growth hormone, also decline with age (Figure 2). The majority of circulating IGF-I is produced in the liver under the control of GH. It appears that the age-related decline in IGF-I production is a direct result of decreases in GH and there is no evidence to suggest increased “GH resistance”. In fact, studies of GH replacement therapy in patients with pituitary disorders and dose-response studies demonstrate a reduction in GH dose necessary to maintain normal IGF-I concentrations in older subjects, although this is due at least in part to the higher susceptibility to side effects from GH and also that their target IGF-1 is lower (6-7).
POTENTIAL MECHANISMS UNDERLYING THE DECLINE IN GH SECRETION WITH AGE
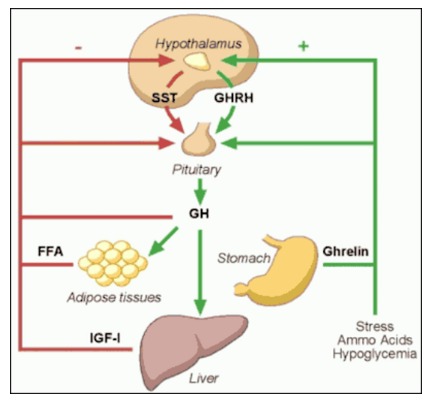
Three hypothalamic factors regulate GH secretion: somatostatin (SRIF), growth-hormone releasing hormone (GHRH) and ghrelin (9) (Figure 3). Somatostatin is a noncompetitive inhibitor of GH secretion as well as of other hormones. It modulates the GH response to GHRH. GHRH is the principal stimulator for GH synthesis and release. Ghrelin is the endogenous ligand to the growth hormone secretagogue receptor-1a (GHSR-1a). Ghrelin is secreted primarily by the stomach and has appetite-stimulating activity separate from its effect on GH secretion. Although recent preclinical data suggest that not all the effects of ghrelin are mediated through GHSR-1a (10), its orexigenic and GH secretagogue effects require the presence of the GHSR-1a (11).Acylated Ghrelin levels decrease with age (12) Growth hormone secretagogues (GHS) are synthetic molecules that stimulate the GHSR-1a exhibiting strong growth hormone-releasing activity.
A variety of stimuli and inhibitors, such as exercise, sleep, food intake, stress and body composition have effects on the hypothalamic factors that regulate GH production (13). All of these factors interact to generate the physiological patterns of pulsatile GH secretion.
There are several mechanisms that could explain the age-related decrease in GH secretion. Possibilities include decreased secretion of GHRH or ghrelin, increase in inhibition by somatostatin, increased sensitivity of somatotrophs to negative feedback inhibition by IGF-I, decline in pituitary responsiveness to GHRH, and pituitary and/or hypothalamic responsiveness to ghrelin.
Whether the aging pituitary responds normally to GHRH and ghrelin is still a matter of debate. Although earlier studies suggest no age–related decline in GH responsiveness to GHRH (13), more recent reports suggest a gender-independent, age-related decline in the GH responsiveness to GHRH and ghrelin (14) (Figure 4).There is no age-related increase in GH sensitivity to IGF-1 (15); however, there may be relative deficiency in GHRH and ghrelin secretion, and an increase in SRIF secretion, in older individuals (16). The density of GHRS-1a receptors in the hypothalamus decreases with aging and this is thought to be responsible for the age-related decreased response to some GHS (17). The aging pituitary is also less responsive to exercise, sleep, and other physiologic stimuli. Based on these observations it is most likely that the age-related change in GH secretion is multifactorial in etiology and is caused by changes above the level of the pituitary.
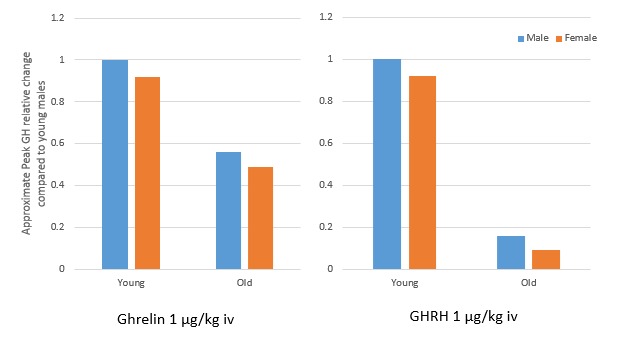
FIGURE 4.
Approximate peak GH relative change in response to I.V. bolus of Ghrelin and GHRH is diminished in older adults compared to young males. Adapted from Broglio F et al. (14).
DECREASE IN GH IN NORMAL AGING: SIMILARITIES WITH AND DIFFERENCES FROM ADULT GROWTH HORMONE DEFICIENCY
Although not a perfect parallel with aging, adult growth hormone deficiency (AGHD) is the best documented source of information on signs and symptoms of reduced GH secretion, effects of treatment, dosing strategies appropriate for adults, and side effects and safety of GH replacement.
Several aspects of normal aging resemble features of the AGHD syndrome, including decrease in muscle and bone mass, increased visceral fat, diminishing exercise and cardiac capacity, atherogenic alterations in lipid profile, thinning of skin, and many psychological and cognitive problems (18-19) (Table 1). Although these changes and the GH deficit of aging are milder than seen in AGHD, they remain clinically significant (20).
Table 1.
Features of Adult Growth Hormone Deficiency (3)
It is important to distinguish the normal decrease in GH secretion associated with aging from true AGHD. Although aging is a state of relative physiologic GH deficiency, it is not a disease in itself and is clearly a separate entity from AGHD. This is demonstrated by higher GH secretion and physiological responses seen in older adults when compared with AGHD patients of similar age (2, 20). Moreover, aging per-se is not an indication for AGHD diagnosis testing or administration (21, 22).
Biochemical tests for AGHD diagnosis are imperfect, and their accuracy is strongly affected by the pre-test probability of the condition. Therefore, the most important indicator of the likelihood of GHD is the clinical context (21). In the majority of cases this is due to tumors arising in the region of the sella turcica or the treatment for these tumors including surgery and radiation, but there are other etiologies. Traumatic brain injury is an increasingly recognized cause of AGHD and may occur without coexisting deficiencies in other anterior pituitary hormones (22). IGF-1 levels alone are generally not enough for AGHD diagnosis, hence the need for provocative testing with the insulin tolerance test, glucagon stimulation test, or when available the combined GHRH-arginine test (23).
The GHS and ghrelin mimetic, macimorelin, has recently been approved by the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for AGHD diagnosis., Macimorelin provides a simple, orally-available, well-tolerated, reproducible, and safe diagnostic test for AGHD with comparable accuracy to the insulin tolerance test in adults (24). Since studies of macimorelin excluded individuals over the age of 65 or with a BMI >40, the safety and efficacy of this test in these populations has not been established.
CLINICAL CONSEQUENCES OF AGE-RELATED DECREASE IN GH SECRETION
The age-related increase in body mass index, changes in body composition, and diminished functional capacity parallel the age-associated decline in growth hormone secretion (18, 19). The alterations in body composition that are most pronounced in normal aging include a reduction in bone density and in muscle mass and strength, an increase in body fat, and adverse changes in lipoprotein profiles (2, 16). This decline in GH production is initially clinically silent, but over time may contribute to sarcopenic obesity and frailty.
The decline in GH may also play a role in cognitive changes observed with aging. One of the many systems for classifying different cognitive domains is grouping them as either “crystallized” or “fluid” intelligence. Crystallized intelligence generally refers to vocabulary and long-term memory; whereas the fluid intelligence includes short term memory and active problem-solving and demonstrates a more marked age-related decline. Several studies have shown a correlation between plasma IGF-I concentrations and performance on tests of fluid intelligence (25), suggesting that GH may play a role in maintenance of fluid intelligence.
Mechanistic insights into the role of growth hormone and IGF-I in age-related alterations in cognitive function were assessed in several studies by Sonntag and colleagues demonstrating somatotrophic effects on rodent brain aging (26). These studies suggest that deficiencies in GH and IGF-I contribute to the functional decline in senescent rats whereas augmentation of GH or IGF-I improved cognitive function, increased glucose utilization throughout the brain, increased cortical vascularity, and ameliorated age-related decline in hippocampal neurogenesis. Although some small studies suggest a positive effect of GHRH or GH replacement on cognition (27), there is insufficient data to recommend GH deficiency testing or GH treatment solely for this purpose (22).
GROWTH HORMONE IN AGING: CAVEATS REGARDING LONGEVITY
Animal studies have called into question the hypothesis that interventions aiming to increase growth hormone secretion and IGF-I should be considered a net benefit (28). In numerous species from nematodes to rodents, caloric restriction, which lowers IGF-1, has been associated with an increased life span. Mice with growth hormone resistance and profoundly reduced IGF-I levels appear healthy and have increased life expectancy, along with reduced fasting glucose and insulin levels (29, 30). In mice with mutations in the gene necessary for the differentiation of pituitary cells to produce GH, life span increases by 42% and tumor development is delayed (30). These findings are accentuated when combined with caloric restriction, while treatment with GH actually reduces lifespan. Mice treated with a metalloproteinase that cleaves an IGF-binding protein to decrease the bioavailability of IGF-1 have a 38% longer lifespan and a lower incidence of tumors. In experiments conducted with a mouse strain prone to developing age-related cognitive decline and decreased life expectancy, treatment with a GHRH-receptor antagonist resulted in increased telomerase activity, improvements in some markers of oxidative stress, improved cognition, and increased mean life expectancy (31) On the other hand, mice treated with a growth hormone antagonist made by a single amino acid substitution in GH do not show increased lifespan.
The mechanism of increased longevity seen in these mice populations is complicated. Some mouse models with increased lifespan have insulin resistance, while those that develop overt diabetes have a shortened lifespan. Long living mice are either leaner than normal or have increased subcutaneous adipose tissue, both of which may have protective aging effects. Caloric restriction and decreased GH/IGF-1 signaling improves resistance to cellular stress, inhibits mTOR by rapamycin, leading to longevity and may be responsible for enhanced tumor resistance.
The parallel between these results and human senescence is not clear. This last assertion is underscored by the recent report that longevity is not increased, but rather reduced in women in a Brazilian population with a GHRH receptor mutation (32) and in a Swiss cohort of patients with isolated GH deficiency from a homozygous mutation spanning the GH1 gene (33). In an Ecuadorian-kindred with GH receptor deficiency and very low levels of IGF-I, however, rates of cancer and diabetes were markedly reduced compared to unaffected people in the same communities (34). A group of Croatian patients with dwarfism and deficiencies in GH, TSH, prolactin, FSH, and LH as a result of a homozygous PROP1 mutation do not die prematurely, do not develop diabetes mellitus, and have delayed appearance of grey hair (30). Ultimately, although size and lifespan appear to have an inverse relationship in some animal studies, no formal longevity studies have been performed in humans with dwarfism.
GROWTH HORMONE THERAPY IN NORMAL AGING
A large literature of over 2000 published papers has led to a general consensus that GH replacement can reverse many abnormalities in AGHD patients. Recent reviews of this literature report reduced fat mass, increased lean body mass, improved exercise capacity and cardiac function, improved bone mineral density, and enhanced quality of life by subjective and objective measures (35-37).
The similarities between aging and adult GH deficiency, while not exact, have led to interest in administering GH directly or stimulating GH secretion in aging individuals. However, the starting point and the target are not the same in the two conditions, and we cannot assume safety and efficacy will be the same.
To date, studies of interventions to increase GH effect in elderly subjects include administration of GH, IGF-I, GHRH, and ghrelin mimetic (GHS) either alone or in combination with each other, sex steroids, or exercise. The first studies of GH treatment in non-GHD older adults were performed not long after its effects in AGHD were demonstrated. In 1990 Rudman and colleagues reported that healthy men above the age of 60 who were treated with GH for 6 months responded with an 8.8% increase in lean body mass, a 14.4% decrease in adipose tissue mass, and a 1.6% increase bone mineral density (BMD) only at the vertebral spine (38). Although the change in BMD was quite small it was especially remarkable considering that most studies of AGHD have required one year or more of therapy to show an improvement in bone density. The changes in body composition persisted after one year of growth hormone treatment (39).
Although the Rudman study did not include any functional measures, given these results, it was postulated that GH treatment might also improve muscle strength and functional performance. Despite an absence of demonstrated functional efficacy, some clinics began to prescribe GH treatment to healthy older persons. Acknowledging this growing practice and the lack of information on the subject, the NIH National Institute on Aging issued a call for applications in 1991 to study trophic factors in aging. Several studies of GH, either alone or in combination with sex steroids, IGF-I, or exercise conditioning, and one study of GHRH were funded and have since been completed. These reports generally demonstrate that GH replacement in normal seniors can increase levels of IGF-I to the young adult normal range. However, lower doses of GH were used in subsequent studies to maintain IGF-I levels in the normal range for healthy young adults, since attempts to reproduce the doses of the initial study of Rudman and colleagues led to severe side effects. Following is a summary of the findings of these studies.
Effects of GH Treatment on Strength and Functional Performance
Though GH treatment of otherwise healthy seniors has been shown to have potentially beneficial effects on body composition, studies of physical functional effects have been generally disappointing and inconsistent.
In a relatively large study, comparing the effects of 6 months of growth hormone treatment with placebo in men aged 70 to 85, Papadakis and colleagues reported a 13% reduction in fat mass and a 4% increase in lean body mass in the treatment group, effects consistent with earlier studies; however, there was no effect of growth hormone on knee or handgrip strength or endurance (40). It is important to point out that this seemingly negative result is likely undercut and confounded by the excellent baseline functional status of the study subjects, who were close to the top of the range on many of the tests used, even before treatment. It is very likely that such a "ceiling effect" led to difficulties in demonstrating further improvements due to treatment with growth hormone.
In a separate study, Taaffe and co-workers showed that exercise training improved strength and exercise capacity, but growth hormone treatment did not further augment this effect (41). Several other similar studies have since been completed (42). These studies were conducted for 6–12 months, each at a single site; therefore, only short-term outcomes and side effects, not long-term risks, could be observed. Their results do not provide guidance on the effects of GH on long-term clinical outcomes or “hard” endpoints such as falls or fractures, maintenance of functional status, or effects on cardiovascular morbidity and mortality – outcomes that could establish more definitively the rationale for GH treatment in normal aging. Though few long-term risks have been observed, this is mainly indicative of an absence of information rather than a demonstration of safety. In 2004 a review of various interventions for sarcopenia and muscle weakness in the elderly concluded that GH therapy produces a high incidence of side-effects, does not increase strength, and that resistance training is the most effective intervention for increasing muscle mass and strength in the elderly (43).
In 2007 Liu and colleagues published a systematic review of the safety and efficacy of growth hormone in the healthy elderly (42). After a mean treatment duration of 27 weeks, GH treated individuals had decrease in fat mass of 2.1 kg and an equal increase in lean body mass of 2.1 kg, with no change in weight overall. Total cholesterol levels trended downward (0.29 mmol/L), though not significantly after adjustment for the change in body composition. Other outcomes, including bone density and other serum lipid levels, did not change. Despite higher doses of GH per kilogram of body weight, women treated with GH did not increase lean body mass and achieved only borderline significant decreases in fat mass, indicating a difference in response to GH therapy between genders. Persons treated with GH were significantly more likely to experience soft tissue edema, arthralgias, carpal tunnel syndrome, and gynecomastia and were somewhat more likely to experience the onset of diabetes mellitus and impaired fasting glucose (Figure 5).
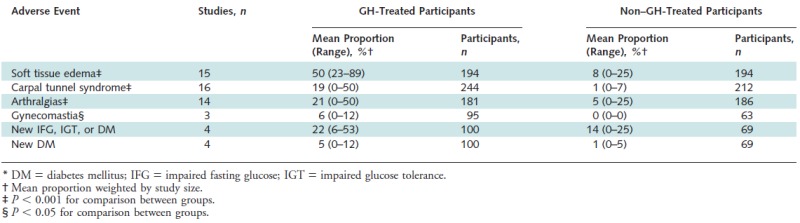
Figure 5.
Adverse events in participants treated with growth hormone versus those not treated. From Liu H et al. (42)
Effects of GH Treatment on Cognition, Sleep, and Mood
As noted above, rodent studies have shown that GH administration increases brain vascularity and improves performance on some cognitive tests, but systematic tests of cognitive effects of GH treatment in humans are lacking (44). A seemingly contradictory finding was reported in 2017 by Basu and colleagues who demonstrated that spatial learning and memory were improved in 12-month-old GH receptor antagonist transgenic mice when compared to their wild type controls, proposing that GH antagonism as well may have cognitive benefits in aging rodents (45). A trial of GH therapy in patients with Down syndrome showed an increase in head circumference but no effect on cognitive performance (46). Early reports suggested that GH increased deep sleep (SWS), but subsequent studies have failed to confirm this and indeed have found increased sleep fragmentation and reduced total deep sleep (27). While GH treatment of adult GH deficiency improves self-rated quality of life (QoL) scores using any of a number of questionnaires, there are no solid comparable data for normal aging.
Combination Interventions with GH and Sex Steroids
In a 6-month study of healthy men and women over the age of 65 using GH alone or in combination with estrogen/progestin in women and testosterone in men, GH administration increased lean body mass (LBM) in women with or without estrogen/progestin (47). In men, GH and testosterone increased LBM when given alone and had an additive effect in combination. In men, total fat mass decreased with either testosterone or GH alone, but the decrease was greatest with the combination, whereas in women GH decreased fat mass while sex steroids did not change fat mass. Body strength did not improve in women and slightly increased in men only in the GH + testosterone arm of the trial. There was no evidence that co-administration of sex steroids altered the frequency or severity of GH-related side effects.
A 2006 British study randomized healthy older men to 6 months treatment with GH, testosterone patch (Te), or combination of both GH and testosterone (GHTe) and compared results to placebo (48). Both GH treated groups experienced similar increases in lean body mass, while this parameter was unchanged by testosterone treatment alone. Fat mass decreased only in the GH/testosterone combination group. Similarly, mid-thigh muscle cross-sectional area and exercise capacity (VO2 max) was increased only in GHTe and not in the GH or Te groups. There was no difference among the groups in 5 of 6 muscle strength measures except for strength of knee flexion that was found to be increased in the GHTe group. Both GH treated groups reported improvement in a quality of life questionnaire. Overall GH treatment was well tolerated in this study, with most GH-related side effects resolving with dose adjustment.
A study published in 2009 randomized men over the age of 65 having IGF-I levels in the bottom tertile of the reference range, and who were treated with testosterone after a “Leydig cell clamp”, to three groups of daily doses of GH either 0, 5 mcg/kg or 10 mcg/kg (49). After 16 weeks the investigators were able to demonstrate significant synergistic effects of GH treatment, when added to testosterone replacement, on all parameters studied including decrease in total fat mass and truncal fat as well as increase in lean body mass and maximal voluntary muscle strength and aerobic endurance. A slight increase in systolic blood pressure was noted in the study, but did not appear to be related to GH therapy.
Growth Hormone and Exercise
Regular exercise has been shown to increase lean body mass, muscle strength, and aerobic capacity in older men (50). Vigorous exercise acutely stimulates growth hormone secretion, a physiologic response that has been utilized as a screening test for growth hormone deficiency in children.
The growth hormone response to exercise decreases with aging (51). This finding led to speculation that some of the effects of exercise might be mediated via effects on growth hormone and IGF-I. Although exercise stimulates an acute rise in growth hormone secretion, subsequent overnight growth hormone secretion is inhibited (52). In older adults, even intensive exercise does not elevate serum IGF-I level (53). Therefore, the effects of exercise on muscle mass and function seem to be separate from those of growth hormone.
Studies assessing the effects of adding GH to progressive resistance training regimens in older adults have found little to no additional benefit of GH therapy on measures of muscle strength or other measures of muscle composition, but did find that GH therapy led to greater reductions in fat mass than resistance training alone (41, 54, 55).
Adverse Effects of GH Treatment
Side effects observed during clinical trials of growth hormone treatment in normal aging must be taken into consideration in a different way from those in patients treated for adult growth hormone deficiency. The possibility that some of the hormonal changes observed with aging could represent adaptive responses must be considered. Whether increasing growth hormone above the age-appropriate normal range may have as many risks, both acute and delayed, as benefits is a worthwhile hypothesis to examine. The acute side effects of growth hormone are largely hormonal. The most worrisome long-term potential side effect, of special importance in the older population where baseline risk is elevated, is the risk of cancer. Though there is no definitive evidence that GH replacement in AGHD increases the risk of de novo or recurrent malignancy, but several case reports note development of cancer after treatment with GH and because it is a mitogen, the use of GH is contraindicated in active malignancy (35, 36). Nevertheless, GH replacement is not associated with tumor regrowth in AGHD patients with pituitary tumors (56).
Older persons are more sensitive to replacement with GH and more susceptible to the side effects of therapy. The acute side effects are due to the hormonal effects of over-replacement, which can be avoided or relieved with careful dose titration. Patients who are older, heavier, or female are more prone to develop complications (22). Common side effects of GH replacement include fluid retention, with peripheral edema, arthralgia, and carpal tunnel syndrome (Figure 5) Although glucose levels often increase with initiation of GH, these levels generally return toward normal with the improvement in body composition and reduced insulin resistance. However, some studies report persistent elevations in fasting glucose and insulin with chronic GH treatment. Other less frequently reported side effects include headache, tinnitus, and benign intracranial hypertension (22). Hypothyroidism is common in the elderly. GH can accelerate both the clearance of thyroxine and promote its conversion to triiodothyronine, and therefore may have variable effects in hypothyroid patients who are on thyroid hormone replacement.
GROWTH HORMONE RELEASING HORMONE (GHRH) AND GROWTH HORMONE SECRETAGOGUES (GHS)
GHRH and GHS stimulate the secretion of GH. Since most AGHD is caused by pituitary lesions, and these patients, unlike healthy seniors, are unresponsive to GHRH or GHS, there are few studies of treatment with these agents.
Theoretically, treatment with GHRH or GHS should lead to more physiologic GH replacement, leading to a pulsatile rather than prolonged elevation in GH and preserving the ability for negative feedback inhibition of GH by increasing IGF-I. GHRH and GHS effects are influenced by the same factors which modulate endogenous GHRH secretion, such as negative feedback by somatostatin. This normal negative feedback regulation would be expected to result in buffering against overdose. The side effects of GHRH treatment are similar in character to GH treatment but are milder and less frequent. Since the GHS are smaller molecules than GH, and generally resistant to digestive enzymes, they can be administered via the oral, transdermal or nasal routes.
Growth Hormone Releasing Hormone (GHRH)
There are several published trials of GHRH treatment in normal aging (57, 58). Once daily GHRH injections can stimulate increases in GH and IGF-I at least to the lower part of the young adult normal range (57). In a study of 6 months treatment with daily bedtime subcutaneous injections of GHRH(1–29)NH2, alone or in combination with formal exercise conditioning, IGF-I levels increased by 35% (56). Participants had an increase in lean body mass and decrease in body fat (mainly abdominal visceral fat). However, there was no improvement in strength or aerobic fitness with GHRH injections. This study confirmed the benefits of exercise but showed no effect upon IGF-I levels; thus, it appears that GH/GHRH and exercise work through different mechanisms. Subjects receiving GHRH also showed no change in scores on an integrated physical functional performance test of activities of daily living, but there was a significant decline in physical function in the placebo group. This finding, suggesting that GHRH can stabilize if not improve physical function, needs confirmation.
Sleep and cognition were also studied in this GHRH trial, with unexpected results. GHRH failed to improve and may even have impaired deep sleep, despite the rise in IGF-I and pulsatile GH. However, GHRH treatment was associated with improved scores in several domains of fluid (but not crystallized) intelligence – those measures previously found to be correlated with circulating IGF-I levels (25).
A 2006 study of the effects of 6-months daily treatment with sermorelin acetate, a GHRH analogue, on cognitive function of 89 elderly adults found significant improvement on several cognitive assessments, particularly those involving problem solving, psychomotor processing speed, and working memory, but no change on tests reflecting crystallized intelligence (27). Higher GH levels were associated with higher Wechsler Adult Intelligence Scale performance IQ scores, and greater increases in IGF-1 were associated with higher verbal fluency test scores, while gender, estrogen status, and initial cognitive function did not interact with the GHRH effect on cognition.
A 2013 pilot study of 30 elderly adults given a stabilized analogue of GHRH, tesamorelin, versus placebo, used magnetic resonance spectroscopy to examine the effects of inhibitory and excitatory neurotransmitters (60). After 20 weeks GABA levels were increased in all brain regions, N-acetylaspartylglutamate levels were increased in the dorsolateral frontal cortex, and myo-inositol (an osmolyte linked to Alzheimer disease) levels were decreased in the posterior cingulate, with similar results across adults with mild cognitive impairment (MCI) and those with normal cognitive function. Treatment related changes in serum IGF-1 were positively correlated with changes in GABA and negatively correlated with myo-inositol. There was a favorable treatment effect on cognition (p = .03), but no significant associations were observed between treatment-related changes in neurochemical and cognitive outcomes.
The follow-up study of 152 elderly patients on tesamorelin versus placebo included those with amnestic MCI (early stage Alzheimer’s disease) and analyzed executive function, episodic memory, mood, sleep, insulin sensitivity, glucose tolerance, body composition, and IGF-1 levels (61). GHRH had a favorable effect on cognition (P = .002) in both groups. Treatment related increases in IGF-I were associated with higher composite change scores in executive function (p = .03). Visual memory, mood, sleep, hemoglobin A1c, and 2-hour OGTT glucose and insulin responses were not affected in either population, though GHRH treatment was associated with increased fasting plasma insulin levels in adults with MCI. Treatment with GHRH reduced body fat by 7.4% (p < .001) and increased lean muscle mass by 3.7% (p < .001), across both populations. Ultimately though, the clinical significance of these results cannot be assessed as no data was collected regarding functional status.
In a non-controlled 3-month trial of GHRH(1-44)amide in 10 postmenopausal women, increases in both GH and IGF-I levels as well as decreased visceral fat were demonstrated (57). This study also reported improvements from baseline in selected measures of functional performance including timed walking and stair climbing.
Thus, as is the case with GH, studies of treatment of healthy seniors with GHRH have arrived at a consensus on hormonal and body composition effects but inconsistent functional effects. There is a very encouraging but still unconfirmed positive effect on some domains of fluid intelligence.
Ghrelin Mimetics and Growth Hormone Secretagogues (GHS)
Ghrelin, a 28 amino-acid octanoylated peptide, is produced in the stomach and increases before meals and during overnight fasting. Ghrelin acts at both hypothalamic and pituitary levels via mechanisms distinct from GHRH. Ghrelin therefore has different effects from GHRH or GH; subjects often gain body weight, lean and fat mass via a number of GH dependent and independent mechanisms (62). The effects of ghrelin on GH secretion depend in part on the presence of GHRH. If GHRH secretion declines with aging, as is thought to be the case, ghrelin’s effects may be blunted. While the effects of these two GHS differ clinically, they have synergistic effects on GH release, and therefore supplementation of both substances may be more effective than either alone. Nevertheless, ghrelin is more potent than GHRH at eliciting GH secretion (14). Additionally, there are other substances which can enhance GH response to GHS by suppressing somatostatin secretion, including arginine and beta-adrenergic antagonists, which could potentially enhance treatment effects (59).
Several studies have shown short-term effects of GHS on GH secretion, but few studies of their chronic effects in normal aging have been reported. Bowers and colleagues showed that chronic repeated injections or subcutaneous infusions of GH-releasing peptide-2 (GHRP-2) could stimulate and maintain increases in episodic GH secretion and raise IGF-I levels (63).
Results of a one-year double-blind, randomized, placebo-controlled, modified-crossover clinical trial of the Merck orally active ghrelin mimetic MK-677 in healthy high functioning older adults were published in 2008 (64). Daily administration of MK-677 significantly increased growth hormone and IGF-I levels to those of healthy young adults without serious adverse effects. Mean fat-free mass decreased in the placebo group but increased in the MK-677 group. No significant differences were observed in abdominal visceral fat or total fat mass. Body weight increased 0.8 kg in the placebo group and 2.7 kg in the MK-677 group (P = 0.003). Fasting blood glucose level increased an average of 0.3 mmol/L (5 mg/dL) in the MK-677 group (P = 0.015), and insulin sensitivity decreased. The most frequent side effects were an increase in appetite that subsided in a few months and transient, mild lower-extremity edema and muscle pain. Low-density lipoprotein cholesterol levels decreased in the MK-677 group relative to baseline values (change, -0.14 mmol/L) (-5.4 mg/dL) P = 0.026); no differences between groups were observed in total or high-density lipoprotein cholesterol levels. Changes in bone mineral density consistent with increased bone remodeling occurred in MK-677 recipients. Increased fat-free mass did not result in changes in strength or function.
A multicenter trial of the Pfizer investigational oral GHS, capromorelin, in pre-frail older men and women recruited over 300 subjects and was initially planned as a two-year intervention (65). The study was stopped, however, after all subjects had been treated for 6 months and many for 12 months, due to failure to see an increase in percent lean body mass, which was a pre-set non-efficacy termination criterion. Absolute lean body mass did increase significantly, but due to the appetite-stimulating and lipogenic/anti-lipolytic effect of ghrelin mimetics – unforeseen in early 1999 when the study was designed and ghrelin was still unknown – subjects also gained weight (about 1.5 Kg) and this washed out the effect on percent lean body mass. However, even this truncated study is currently the largest clinical trial of chronic GHS treatment in aging. It showed the expected increases in IGF-I levels and (as noted) total lean body mass. There were also encouraging effects on physical functional performance. Of seven functional tests, one improved significantly after 6 months of treatment, and another after 12 months. Two other measures showed non-significant trends toward improvement, and the three remaining measures showed no effect. Effects on clinical endpoints such as falls could not be assessed with this relatively brief duration of treatment. Side effects were generally mild, including increases in fasting blood sugar within the normal range. Interestingly, there was a self-reported deterioration of sleep quality, though formal sleep testing was not performed. Cognition was not studied in this trial. The reasons for the difference in functional outcomes between the two trials are not clear, but it is speculated that this may reflect differences in the populations studied. The MK-677 study recruited a robustly healthy population of seniors in whom further improvement in physical function might be difficult to achieve, while the capromorelin trial was limited to participants already manifesting a decline in function.
Thus, as with GH and GHRH, reports of the hormonal and body composition effects of ghrelin mimetic GHS in normal aging are relatively consistent, but there is no consensus on functional effects among these very few studies, and of course none could assess long-term clinical outcomes or risks.
The novel GHS, anamorelin, is currently under clinical development for cancer anorexia and cachexia syndrome (CACS), a syndrome overrepresented in the elderly. In a phase II randomized, double-blind, placebo-controlled study, 3 days of treatment increased body weight and appetite in these patients when compared to placebo (66). Over 3 months of treatment, anamorelin increased body weight, LBM, hand grip strength, and quality of life (QOL). Anamorelin also increased IGF-1 and IGF binding protein (IGFBP)-3. It was well-tolerated, but it induced a small increase in glucose and insulin concentrations (67). Unfortunately, two large, international, randomized, double-blind, placebo-controlled phase III studies in patients with advanced non-small cell lung cancer and CACS (ROMANA 1 and 2) did not show improvements in handgrip strength with anamorelin, in spite of increased LBM, fat mass, body weight and appetite-related QoL compared to placebo (68). Although these studies were not restricted to the elderly, the mean age of the population was above 60 years of age in all studies.
CONCLUSIONS
While aging is not a disease, it results in alterations in body composition and functional decline with subsequent frailty and loss of independence. Interventions that slow this decline could potentially prolong the capacity for independent living and improve quality of life, but this has not yet been demonstrated. It is unknown whether the decrease in trophic hormones including sex steroids and growth hormone that occur with aging represents an adaptive or pathological process. Aging may represent a milder form of adult GHD, and since GH replacement in frank AGHD has met with success, it may be logical to reason that GH replacement or stimulation by GHRH or GHS might be beneficial in aging. However, older persons are more sensitive to GH, and thus more susceptible to the side effects of replacement. To date, definitive conclusions regarding functional effects of treatments in normal aging aimed at increasing GH levels to those of young healthy persons have been elusive. Until more studies are undertaken to determine the long-term effects of GH and GHS supplementation, conclusive statements about the merits of treatment cannot be made. Long term studies on hard clinical endpoints, such as falls and fracture rates, function measures, quality of life, and decreased morbidity and mortality from vascular disease need to be performed in order to establish the role, if any, for GH and GHS treatment in normal aging. In the meantime, GH use for anti-aging purposes is currently prohibited by US federal law (69, 70).
REFERENCES
- 1.
- Sattler FR. Growth hormone in the aging male. Best Pract Res Clin Endocrinol Metab. 2013;27(4):51–55. [PMC free article: PMC3940699] [PubMed: 24054930]
- 2.
- Anawalt BD, Merriam GR. Neuroendocrine aging in men: andropause and somatopause. Endocrinology and Metabolism Clinics of North America. 2001;30:647–69. [PubMed: 11571935]
- 3.
- Merriam GR, Hersch EC. Growth hormone (GH)-releasing hormone and GH secretagogues in normal aging: Fountain of Youth or Pool of Tantalus? Clin Interv Aging. 2008;3(1):121–9. [PMC free article: PMC2544358] [PubMed: 18488883]
- 4.
- Ho KY, Evans WS, Blizzard RM, Velduis JD, Merriam GR, Samojlik R, Furlanetto R, Rogol AD, Kaiser DL, Thorner MO. Effects of sex and age on the 24 hour profile of growth hormone secretion in man: Importance of endogenous estradiol concentrations. J Clin Endocrinol Metab. 1987;64:51–8. [PubMed: 3782436]
- 5.
- Maheshwari H, Sharma L, Baumann G. Decline of plasma growth hormone binding protein in old age. J Clin Endocrinol Metab. 1996 Mar;81(3):995–7. [PubMed: 8772563]
- 6.
- Jørgensen JOL, Flyvbjerg A, Lauritzen T, Alberti KGMM, Ørskov H, Christiansen JS. Dose-response studies with biosynthetic human growth hormone deficient patients. J Clin Endocrinol Metab. 1988;67:36–40. [PubMed: 3288652]
- 7.
- Møller J, Jørgensen JOL, Laursen T, Frystyk J, Naeraa R, Ørskov H, Christiansen JS. Growth hormone (GH) dose regimens in GH deficiency: effects on biochemical growth markers and metabolic parameters. Clin Endocrinol. 1993;39:403–408. [PubMed: 7507009]
- 8.
- Juul A, Bang P, Hertel NT, Main K, Dalgaard P, Jørgensen K, Müller J, Hall K, Skakkebaek NE. Serum insulin-like growth factor-I in 1030 healthy children, adolescents,and adults: relation to age, sex, stage of puberty, testicular size, and body mass index. J Clin Endocrinol Metab. 1994;78:744–752. [PubMed: 8126152]
- 9.
- Veldhuis JD, Bowers CY. Human GH pulsatility: an ensemble property regulated by age and gender. J Endocrinol Invest. 2003 Sep;26:799–813. [PubMed: 14964431]
- 10.
- Chen JA, Spencer A, Guillory B, Luo J, Mendiratta M, Belinova B, Halder T, Zhang G, Li YP, Garcia JM. Ghrelin prevents tomour- and cisplatin-induced muscle wasting: characterization of multiple mechanisms involved. J Cachexia Sarcopenia Muscle. 2015 Jun;2(6):132–43. [PMC free article: PMC4458079] [PubMed: 26136189]
- 11.
- Sun Y, Wang P, Zheng H, Smith RG. Ghrelin stimulation of growth hormone release and appetite is mediated through the growth hormone secretagogue receptor. Proc Natl Acad Sci USA. 2004 Mar 30;101(13):4670–84. [PMC free article: PMC384806] [PubMed: 15070777]
- 12.
- Di Francesco V, Fantin F, Residori L, Bissoli L, Micciolo R, Zivelonghi A, Zoico E, Omizzolo F, Bosello O, Zamboni M. Effect of age on the dynamics of acylated ghrelin in fasting conditions and in response to a meal. J Am Geriatr Soc. 2008 Jul;56(7):1369–70. [PubMed: 18774977] [CrossRef]
- 13.
- Pavlov EP, Harman SM, Merriam GR, Gelato MC, Blackman MR. Responses of growth hormone (GH) and somatomedin C to GH-releasing hormone in healthy aging men. J Clin Endocrinol Metab. 1986;62:595. [PubMed: 3080468]
- 14.
- Broglio F, Benso A, Castiglioni C, Gottero C, Prodam F, Destefanis S, Guana C, van de Lely AJ, Deghenghi R, Bo M, Arvat E, Ghigo E. The endocrine response to ghrelin as a response to gender in humans in young and elderly subjects. J Clin Endocrinol Metab. 2003 Apr;88(4):1537–42. [PubMed: 12679436]
- 15.
- Chapman IM, Hartman ML, Pezzoli SS, Harrell FE Jr, Hintz RL, Alberti KG, Thorner MO. Effect of aging on the sensitivity of growth hormone secretion to insulin-like growth factor-I negative feedback. J Clin Endocrinol Metab. 1997;82:2996. [PubMed: 9284733]
- 16.
- Russell-Aulet M, Jaffe CA, Demott-Friberg R, Barkan AL. In vivo semiquantification of hypothalamic growth hormone-releasing hormone (GHRH) output in humans: Evidence for relative GHRH deficiency in aging. J Clin Endocrinol Metab. 1999;84:3490. [PubMed: 10522985]
- 17.
- Ghigo E, Arvat E, Giordano R, Broglio F, Gianotti L, Maccario M, Bisi G, Graziani A, Papotti M, Muccioli G, Deghenghi R, Camanni F. Biologic activities of growth hormone secretagogues in humans. Endocrine. 2001 Feb 14;(1):87–93. [PubMed: 11322506]
- 18.
- Corpas E, Harman SM, Blackman MR. Human growth hormone and human aging. Endocr. Rev. 1993;14:20–39. [PubMed: 8491152]
- 19.
- Martin FC, Yeo A-L, Sönksen PH. Growth hormone secretion in the elderly: aging and the somatopause. Balliere’s Clin Endocrinol Metab. 1997;11:223–50. [PubMed: 9403121]
- 20.
- Toogood AA, O’Neill PA, Shalet SM. Beyond the somatopause: growth hormone deficiency in adults over the age of 60 years. J Clin Endocrinol Metab. 1996;81:460–65. [PubMed: 8636250]
- 21.
- h1>Merriam GR, Wyatt FG. Diagnosis and treatment of growth hormone deficiency in adults: current perspectives. Current Opinion in Endocrinology and Diabetes. 2006;13:362–8.
- 22.
- Molitch ME, Clemmons DR, Malozowski S, Merriam GR, Vance ML. Evaluation and treatment of adult growth hormone deficiency: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2011 Jun;96(6):1587–1609. [PubMed: 21602453]
- 23.
- Kargi AY, Merriam GR. Testing for growth hormone deficiency in adults: doing without growth hormone-releasing hormone. Curr Opin Endocrinol Diabetes Obes. 2012;19(4):300–5. [PubMed: 22596248]
- 24.
- Garcia JM, Biller BMK, Korbonits M, Popovic V, Luger A, Strasburger CJ, Chanson P, Medic-Stojanoska M, Schophol J, Zakrzewska A, Pekic S, Bolanowski M, Swerdloff R, Wang C, Blevins T, Marcelli M, Ammer N, Sachse R, Yuen KCJ. Macimorelin as a diagnostic test for adult GH deficiency. J Clin Endocrinol Metab. 2018 Aug 1;103(8):3083–3093. [PubMed: 29860473]
- 25.
- Aleman A, Verhaar HJ, DeHaan EH, De Vries WR, Samson MM, Drent ML, Van de Veen EA, Koppeschaar HP. Insulin-like growth factor-I and cognitive function in healthy older men. J Clin Endocrinol Metab. 1999;84:471–5. [PubMed: 10022403]
- 26.
- Sonntag WE, Ramsey M, Carter CS. Growth hormone and insulin-like growth factor-1 (IGF-1) and their influence on cognitive aging. Aging Res Rev.h1>2005; 4(2):195-212. [PubMed: 16024298]
- 27.
- Vitiello MV, Moe KE, Merriam GR, Mazzoni G, Buchner DH, Schwartz RS. Growth hormone releasing hormone improves the cognition of healthy older adults. Neurobiol Aging.h1>2006; 27:318-23. [PubMed: 16399214]
- 28.
- Bartke A. Growth hormone and aging: updated review. World J Mens Health. 2019 Jan;37(1):19–30. [PMC free article: PMC6305861] [PubMed: 29756419]
- 29.
- Coschigano KT, Clemmons D, Bellush LL, Kopchick JJ. Assessment of growth parameters and life span of GHR/BP gene-disrupted mice. Endocrinology. 2000;141:2608. [PubMed: 10875265]
- 30.
- Junnila RK, List EO, Berryman DE, Murrey JW, Kopchick JJ. The GH/IGF-1 axis in ageing and longevity. Nat Rev Endocrinol. 2013;9:366–376. [PMC free article: PMC4074016] [PubMed: 23591370]
- 31.
- Banks WA, Morley JE, Farr SA, Price TO, Ercal N, Vidaurre I, Schally AV. Effects of a growth hormone-releasing hormone antagonist on telomerase activity, oxidative stress, longevity, and aging in mice. Proc Natl Acad Sci U S A. 2010 Dec 21;107(51):22272–7. [PMC free article: PMC3009756] [PubMed: 21135231]
- 32.
- Aguiar-Oliveira MH, Oliveira FT, Pereira RM, Oliveira CR, Blackford A, Valenca EH, Santos EG, Gois-Junior MB, Meneguz-Moreno RA, Araugo VP, Oliveira-Neto LA, Almeida RP, Santos MA, Farias NT, Silveira DC, Cabral GW, Calazans FR, Seabra JD, Lopes TF, Rodrigues EO, Porto LA, Oliveira IP, Melo EV, Martari M, Salvatori R. Longevity in untreated congenital growth hormone deficiency due to a homozygous mutation in the GHRH receptor gene. J Clin Endocrinol Metab. 2010;95:714–21. [PMC free article: PMC2840870] [PubMed: 19965916]
- 33.
- Besson AS, Gallati S, Jenal A, Horn R, Mullis PS, Mullis PE. Reduced longevity in untreated patients with isolated growth hormone deficiency. J Clin Endocrinol Metab. 2003;88(8):3664–7. [PubMed: 12915652]
- 34.
- Guevara-Aguirre J, Balasubramanian P, Guevara-Aguirre M, Wei M, Madia F, Cheng CW, Hwang D, Martin-Montalvo A, Saavedra J, Ingles S, de Cabo R, Cohen P, Longo VD. Growth hormone receptor deficiency is associated with a major reduction in pro-aging signaling, cancer, and diabetes in humans. Sci Transl Med.h1>2011; 3: 70 ra13. [PMC free article: PMC3357623] [PubMed: 21325617]
- 35.
- Reed ML, Merriam GR, Kargi AY. Adult growth hormone deficiency - benefits, side effects, and risks of growth hormone replacement. Front Endocrinol. 2013;4:64. [PMC free article: PMC3671347] [PubMed: 23761782]
- 36.
- Kargi AY, Merriam GR. Diagnosis and treatment of growth hormone deficiency in adults. Nature Reviews Endocrinology. 2013;9:335–345. [PubMed: 23629539]
- 37.
- Melmed S. Pathogenesis and diagnosis of growth hormone deficiency in adults. N Engl J Med. 2019 Jun 27;380(26):2551–2562. [PubMed: 31242363]
- 38.
- Rudman D, Feller AG, Nagraj HS, Gergans GA, Lalitha PY, Goldberg AF, Schlenker RA, Cohn L, Rudman IW, Mattson DE. Effects of human growth hormone in men over 60 years old. N Engl J Med. 1990;323:1–6. [PubMed: 2355952]
- 39.
- Rudman D, Feller AG, Cohn L, Shetty KR, Rudman IW, Draper MW. Effects of human growth hormone on body composition in elderly men. Horm Res. 1991;36 suppl:73. [PubMed: 1806490]
- 40.
- Papadakis MA, Grady D, Black D, Tierney MJ, Gooding GA, Schambelan M, Grunfeld C. Growth hormone replacement in healthy older men improves body composition but not functional ability. Ann Intern Med. 1996;124:708. [PubMed: 8633830]
- 41.
- Taaffe DR, Jin IH, Vu TH, Hoffman AR, Marcus R. Lack of effect of recombinant human growth hormone on muscle morphology and GH-insulin-like growth factor expression in resistance-trained elderly men. J Clin Endocrinol Metab. 1996;81:421. [PubMed: 8550787]
- 42.
- Liu H, Bravata DM, Olkin I, Nayak S, Roberts B, Garber AM, Hoffman AR. Systematic review: the safety and efficacy of growth hormone in the healthy elderly. Ann Int Med. 2007;146:104–115. [PubMed: 17227934]
- 43.
- Borst SE. Interventions for sarcopenia and muscle weakness in older people. Age Ageing. 2004;33(6):548–555. [PubMed: 15385272]
- 44.
- Ashpole NM, Sanders JE, Hodges EL, Yan H, Sonntag WE. Growth hormone, insulin-like growth factor-1 and the aging brain. Exp Gerontol. 2014; S0531-5565(14)00277-0 E-pub. [PMC free article: PMC4388761] [PubMed: 25300732]
- 45.
- Basu A, McFarlane HG, Kopchick JJ. Spatial learning and memory in male mice with altered growth hormone action. Hormone Behav. 2017;93:18–30. [PubMed: 28389277]
- 46.
- Growth Hormone Treatment in Down's Syndrome, eds. S.Castells and K.E.Wisniewski, London, J.Wiley,1993).
- 47.
- Blackman MR, Sorkin JD, Munzer T, Bellantoni MF, Busby-Whitehead J, Stevens TE, Jayme J, O’Connor KG, Christmas C, Tobin JD, Stewart JK, Cottrell E, St Clair C, Pabst KM, Harman SM. Growth hormone and sex steroid administration in healthy aged women and men: a randomized controlled trial. JAMA. 2002;288:2282–92. [PubMed: 12425705]
- 48.
- Giannoulis MG, Sonsken PH, Umpleby M, Breen L, Pentecost C, Whyte M, McMillan CV, Bradley C, Martin FC. The effects of growth hormone and/or testosterone in healthy elderly men: a randomized controlled trial. J Clin Endocrinol Metab. 2006 Feb;91(2):477–84. [PubMed: 16332938]
- 49.
- Sattler FR, Castaneda-Sceppa C, Binder EF, Schroeder ET, Wang Y, Bhasin S, Kawakubo M, Stewart Y, Yarasheski KE, Ulloor J, Colletti P, Roubenoff R, Azen SP. Testosterone and growth hormone improve body composition and muscle performance in older men. J Clin Endocrinol Metab. 2009;94(6):1991–2001. [PMC free article: PMC2690426] [PubMed: 19293261]
- 50.
- Pate RR, Pratt M, Blair SN, Haskell WL, Macera CA, Bouchard C, Buchner D, Ettinger W, Heath GW, King AC. Physical activity and public health: A recommendation from the Centers for Disease Control and Prevention and the American College of Sports Medicine. JAMA. 1995;273:402. [PubMed: 7823386]
- 51.
- Kraemer WJ, Hakkinen K, Newton RU, Nindl BC, Volek JS, McCormick M, Gotshalk LA, Gordon SE, Fleck SJ, Campbell WW, Putukian M, Evans WJ. Effects of heavy-resistance training on hormonal response patterns in younger vs. older men. J Appl Physiol. 1999;87:982. [PubMed: 10484567]
- 52.
- Nindl BC, Hymer WC, Deaver DR, Kraemer WJ. Growth hormone pulsatility profile characteristics following acute heavy resistance exercise. J Appl Physiol. 2001;91(1):163–72. [PubMed: 11408427]
- 53.
- Vitiello MV, Wilkinson CW, Merriam GR, Moe KE, Prinz PN, Ralph DD, Colasurdo EA, Schwartz RS. Successful 6-month endurance training does not alter insulin-like growth factor-I in healthy older men and women. J Gerontol Med Sci. 1997;52A:149–154. [PubMed: 9158556]
- 54.
- Yarasheski KE, Zachwieja JJ, Campbell JA, et al. Effect of growth hormone and resistance exercise on muscle growth and strength in older men. American Journal of Physiology. 1995;268(2 Pt 1):E268–E276. [PubMed: 7864103]
- 55.
- Hennessey JV, Chromiak JA, DellaVentura S, et al. Growth hormone administration and exercise effects on muscle fiber type and diameter in moderately frail older people. Journal of the American Geriatrics Society. 2001;49(7):852–858. [PubMed: 11527474]
- 56.
- Arnold JR, Arnold DF, Marland A, Karavitaki N, Wass JA. Clin Endocrinol (Oxf). 2009;70:435–438. GH replacement in patients with non-functioning pituitary adenoma (NFA) treated solely by surgery is not associated with increased risk of tumour recurrence. [PubMed: 19236640]
- 57.
- Veldhuis JD, Patri JM, Frick K, Weltman JY, Weltman AL. Administration of recombinant human GHRH-1,44-amide for 3 months reduces abdominal visceral fat mass and increases physical performance measures in postmenopausal women. Eur J Endocrinol. 2005;153:669–77. [PubMed: 16260425]
- 58.
- Merriam GR, Kletke M, Barsness S, Buchner D, Hirth V, Moe KE, Schwartz RS, Vitiello MV. Growth hormone-releasing hormone in normal aging: An Update. Today’s Therapeutic Trends. 2000;18:335–54.
- 59.
- Merriam GR, Buchner DM, Prinz PN, Schwartz RS, Vitiello MV. Potential applications of GH secretagogs in the evaluation and treatment of the age-related decline in growth hormone secretion. Endocrine. 1997;7:1–3. [PubMed: 9449031]
- 60.
- Friedman SD, Baker LD, Borson S, Jensen JE, Barsness SM, Craft S, Merriam GR, Otto RK, Novotny EJ, Vitiello MV. Growth hormone-releasing hormone effects on brain γ-aminobutyric acid levels in mild cognitive impairment and healthy aging. JAMA Neurol. 2013;70(7):883–890. [PMC free article: PMC3764915] [PubMed: 23689947]
- 61.
- Baker LD, Barsness SM, Borson S, Merriam GR, Friedman SD, Craft S, Vitiello MV. Effects of growth hormone-releasing hormone on cognitive function in adults with mild cognitive impairment and healthy older adults: Results of a controlled trial. Arch Neurol. 2012;69(11):1420–1429. [PMC free article: PMC3764914] [PubMed: 22869065]
- 62.
- Guillory B, Splenser A, Garcia J. The role of ghrelin in anorexia-cachexia syndromes. Vitam Horm. 2013;92:61–106. [PubMed: 23601421]
- 63.
- Bowers CY, Granda R, Mohan S, Kuipers J, Baylink D, Veldhuis JD. Sustained elevation of pulsatile growth hormone (GH) secretion and insulin-like growth factor I (IGF-I), IGF-binding protein-3 (IGFBP-3), and IGFBP-5 concentrations during 30-day continuous subcutaneous infusion of GH-releasing peptide-2 in older men and women. J Clin Endocrinol Metab. 2004;89:2290–300. [PubMed: 15126555]
- 64.
- Nass R, Pezzoli SS, Oliveri MC, Patrie JT, Harrell FE Jr, Clasey JL, Heymsfield SB, Bach MA, Vance ML, Thorner MO. Effects of an oral ghrelin mimetic on body composition and clinical outcomes in healthy older adults: a randomized trial. Ann Intern Med.h1>2008;149 (9):601-11. [PMC free article: PMC2757071] [PubMed: 18981485]
- 65.
- White HK, Petrie CD, Landschulz W, MacLean D, Taylor A, Lyles K, Wei JY, Hoffman AR, Salvatori R, Ettinger MP, Morey MC, Blackman MR, Merriam GR., Capromorelin Study Group. Effects of an oral growth hormone secretagogue in older adults. J Clin Endocrinol Metab. 2009;94(4):1198–206. [PubMed: 19174493]
- 66.
- Garcia JM, Friend J, Allen S. Therapeutic potential of anamorelin, a novel, oral ghrelin mimetic, in patients with cancer-related cachexia: A multicenter, randomized, double-blind, crossover, pilot study. Support Care Cancer. 2013;21(1):129–13. [PubMed: 22699302]
- 67.
- Garcia JM, Boccia RV, Graham CD, Yan Y, Duus EM, Allen S, Friend J. Anamorelin for patients with cancer cachexia: An integrated analysis of two phase 2, randomised, placebo-controlled, double-blind trials. Lancet Oncol. 2015;16(1):108–116. [PubMed: 25524795]
- 68.
- Temel JS, Abernethy AP, Currow DC, Friend J, Duus EM, Yan Y, Fearon KC. Anamorelin in patients with non-small-cell lung cancer and cachexia (romana 1 and romana 2): Results from two randomised, double-blind, phase 3 trials. Lancet Oncol. 2016;17(4):519–531. Phase III clinical trial results of anamorelin in CACS. [PubMed: 26906526]
- 69.
- Perls TT, Reisman NR, Olshansky SJ. Provision or distribution of growth hormone for “antiaging” clinical and legal issues. JAMA. 2005;294(16):2086–2090. [PubMed: 16249424]
- 70.
- Sonksen P. Idiopathic growth hormone deficiency in adults, Ben Johnson, and the somatopause. J Clin Endocrinol Metab. 2013 Jun;98(6):2270–3. [PubMed: 23729014]
Publication Details
Author Information and Affiliations
Publication History
Last Update: October 7, 2019.
Copyright
This electronic version has been made freely available under a Creative Commons (CC-BY-NC-ND) license. A copy of the license can be viewed at http://creativecommons.org/licenses/by-nc-nd/2.0/.
Publisher
MDText.com, Inc., South Dartmouth (MA)
NLM Citation
Garcia JM, Merriam GR, Kargi AY. Growth Hormone in Aging. [Updated 2019 Oct 7]. In: Feingold KR, Anawalt B, Blackman MR, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-.