NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Feingold KR, Anawalt B, Blackman MR, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-.
ABSTRACT
Pancreatic neuroendocrine neoplasms (PNENs) are a heterogenous group of relatively rare pancreatic malignancies with a unique biology and pathophysiology. Over the last few years, there have been significant improvements in imaging and treatment strategies, which have led to advances in patient’s management and quality of life (QOL). Yet, in practice, there are still a number of unanswered questions. For example, it remains a challenge to choose the optimal treatment sequence from the plethora of options and to properly monitor PNEN patients. Therefore, in this chapter, recent advances in the pathophysiology, diagnosis, monitoring, and management of these neoplasms will be summarized and placed in a historical context. For complete coverage of all related areas of Endocrinology, please visit our on-line FREE web-text, WWW.ENDOTEXT.ORG.
INTRODUCTION
Pancreatic neuroendocrine neoplasms (PNENs) are an uncommon subset of neuroendocrine neoplasms (NENs) originating from endocrine cells (1-3). PNENs represent 1-2% of all pancreatic neoplasms and according to the Surveillance, Epidemiology and End Results (SEER) program, the annual age-adjusted incidence has risen from 0.32/100,000 persons in 2004 to 0.48/100,000 persons in 2021 (2, 4-7). Improvements in and a wider availability of high-quality imaging techniques and a well-established classification system are believed to be major factors in the increasing incidence of PNENs (5, 8, 9).
PNENs can be divided into both functional (10-40%) and non-functional (60-90%) neoplasms (2, 6, 7, 10, 11). Functional PNENs (F-PNENs) are characterized by a specific clinical course and symptoms due to excessive hormone production (e.g., insulin, gastrin) (10-12). The most frequent, recognized F-PNENs are listed in Table 1 (1). Less common F-PNENs include somatostatinomas, ACTHomas and PNENs that cause carcinoid syndrome, acromegaly, or hypercalcemia (2). Patients with non-functional PNENs (NF-PNENs) lack symptoms related to clinical hormonal syndromes and are therefore usually diagnosed at a more advanced stage with characteristically large primary tumors (70% >5 cm) and liver metastasis in more than 60% of the cases (2, 9, 12, 13). NF-PNENs are hence frequently discovered by chance on imaging studies performed due to nonspecific abdominal pain, often caused by tumor bulk (2, 9, 12, 14). Although NF-PNENs do not secrete peptides causing clinical syndromes, they characteristically secrete a number of other peptides including chromogranin A (CgA) and pancreatic polypeptide (PP). However, elevated levels of PP or CgA are not specific for NF-PNENs as they are also observed in patients with renal failure and inflammatory conditions (2, 9, 12-14).
Table 1.
Overview of Recognized Functional PNENs and Their Characteristics
| Tumor [syndrome] | Hormone | Clinical symptoms | Biochemical diagnosis |
|---|---|---|---|
| Insulinoma [Whipple’s triad] | Insulin | Hypoglycemia | At hypoglycemia: Insulin > 6 µU/L Glucose 40 mg/dL C-peptide 0.6 ng/mL Proinsulin ≥ 20 pmol/L |
| Gastrinoma [Zollinger-Ellison] | Gastrin | Abdominal pain, Gastroesophageal reflux, Diarrhea, Duodenal ulcers | Serum fasting gastrin level ≥ 10 times normal range |
| VIPoma [Verner-Morrison] | Vasoactive intestinal peptide (VIP) | Severe watery diarrhea, Hypokalemia | Fasting serum VIP > 60 pmol/L |
| Glucagonoma | Glucagon | Rash, Glucose intolerance (diabetes), Necrolytic migratory erythema, Weight loss | Fasting glucagon > 500 pg/mL |
Note: This table was assembled based on information from Gastroenterology, Metz D. and Jensen R., Gastrointestinal neuroendocrine tumors: Pancreatic Endocrine tumors, 1469-1492 © 2008 (2) and World Journal of Gastroenterology, Ma Z., Gong Y., Zhuang H. et al., Pancreatic neuroendocrine tumors: A review of serum biomarkers, staging and management, 2305-2322 © 2020 (7) and Current Opinion in Gastroenterology, Perri G., Prakash L. and Katz M., Pancreatic neuroendocrine tumors, 468-477 © 2019 (3).
CLASSIFICATION AND STAGING
The World Health Organization (WHO) classification from 2019 (Table 2) takes into account both differentiation status and proliferation rate of the tumor. The former is determined through a histological examination of tumor morphology in which well-differentiated neuroendocrine tumors (NETs) can be distinguished from poorly differentiated neuroendocrine carcinomas (NECs). A grade is then assigned based on the proliferation rate assessed via Ki-67 index and mitotic count. Well-differentiated NETs can be divided into low grade (G1), intermediate grade (G2), and high grade (G3) tumors that have respective Ki-67 values of <3%, 3-20%, and >20% or mitotic counts of <2, 2-20, and >20 per 2mm3 (10 high power fields (HPF)). In the poorly-differentiated NEC group (small and large cell types), only high grade G3 tumors with a Ki-67 value >20 are found. In addition, neoplasms exist that consist of neuroendocrine cells as well as non-neuroendocrine adenocarcinoma or squamous carcinoma cells (i.e., mixed non-neuroendocrine-neuroendocrine neoplasms (MiNEN)) (3, 6-8, 15, 16). Depending on tumor grade and primary site, the 5-year survival varies between 15-95% and median overall survival (OS) from approximately 12 years for patients with G1 to 10 months in patients with G3 PNENs (3, 17). PNENs most often occur sporadically, but can also occur in patients with various inherited disorders (2, 18). For example, PNENs develop in 80-100% of patients with Multiple Endocrine Neoplasia type 1 (MEN1), in 10-17% of patients with von Hippel-Lindau syndrome (VHL), and occasionally in patients with tuberous sclerosis and neurofibromatosis (3, 18).
Table 2.
WHO Classification (2019) of PNENs
| Type | Differentiation status | Grade | Proliferation rate | |
|---|---|---|---|---|
| Ki-67 (%) | Mitotic count (2mm2) | |||
| NEN | Well-differentiated NETs | G1 | < 3 | < 2 |
| G2 | 3 – 20 | 2 – 20 | ||
| G3 | > 20 | > 20 | ||
| Poorly-differentiated NECs Small cell (SCNECs) Large cell (LCNECs) | G3 | > 20 | > 20 | |
| MiNEN | NET or NEC + ADC or SCC | G1-G3 | See above | See above |
Note: This table was adapted from Histopathology, Nagtegaal I., Odze R., Klimstra D. et al., The 2019 WHO classification of tumours of the digestive system, 182-188. © 2019 (16). NEC- neuroendocrine carcinomas; NET- neuroendocrine tumors; ADC- adenocarcinoma cells; SCC- squamous carcinoma cells
PNENs are also classified based on the tumor-node-metastasis (TNM) classification which estimates the prognosis of the tumors based on the anatomy of the tumor (3). Previously there was no generally accepted staging system, so in Europe usually the European Neuroendocrine Tumor Society (ENETS) staging system was applied, while in America the America Joint Committee on Cancer (AJCC)/Union for International Cancer Control (UICC) system was being used (19-21). In the 7th edition of the AJCC/UICC, the same ordering system was employed for PNENs as for pancreatic adenocarcinoma (PAAD), but due to biological differences between both tumor types, this staging system proved to have some limitations (19, 20). Consequently, in the revised 8th edition of the AJCC/UICC, the classification system of ENETS was implemented (21). Two research groups demonstrated that the system employed in this 8th edition was superior to that of the 7th edition as well as the ENETS staging system and should be considered as golden standard (20, 22).
INDUCTION OF PNENS
PNENs are also often referred to as islet cell tumors since it is presumed that they arise from the islets of Langerhans (3, 23, 24). These islets contain A-, B-, D-, D1-, and D2-cells that respectively secrete glucagon, insulin, somatostatin, pancreatic polypeptide, and vasoactive intestinal polypeptide (VIP) (25). Logically, the F-PNENs most definitely arise from these cells, but the cell of origin in NF-PNENs is still a matter of debate (26, 27). Chan et al. revealed that NF-PNENs with ATRX, DAXX, and MEN1 mutations (A-D-M mutant) had a worse clinical outcome than A-D-M wild-type (WT) tumors. In addition, they were able to demonstrate, through RNA sequencing and DNA methylation analysis, that the A-D-M mutant PNENs had high ARX and low PDX1 expression which is consistent with the expression profile found in α-cells (28). Cejas et al. found that NF-PNENs could be divided into two subgroups with epigenomes and transcriptomes very similar to those of α- and β-cells, respectively (29). These findings were confirmed by Di Domenico and colleagues who were able to demonstrate that the genome-wide DNA methylation profiles of NF-PNENs were very consistent with the methylation profiles of α- and β-cells (24). Based on these findings, it was hypothesized that NF-PNENs evolve primarily but not exclusively from the α-cell lineage and β-cell lineage (27).
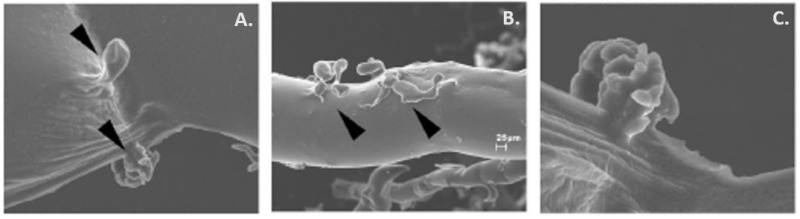
Figure 1.
Visualization of the pancreatic duct glandular structures (PDGs) (arrowheads) in (A) large and (B) small ducts using scanning electron microscopy (SEM). PDGs can occur as single outpouches or form a complex of sac-like dilatations as illustrated in (C). This figure has been adapted from Gastroenterology, Strobel O., Rosow D. E., Rakhlin E. Y., et al., Pancreatic duct glands are distinct ductal compartments that react to chronic injury and mediate Shh-induced metaplasia, 138 (3): 1166-77 © 2010 (30).
Others in turn suggest that PNENs develop from multipotent pancreatic progenitor (MPP) cells in the ductal and islet regions of the pancreas that would be able to generate new pancreatic islet cells (31, 32). However, it remains unclear whether these cells originate in the islets or whether they migrate from the pancreatic ducts to subsequently transform into endocrine cells (33). This hypothesis is strengthened by the fact that early endocrine progenitors in fact appear to originate from a bipotent ductal endocrine progenitor, which in turn originates from MPP cells (34). However, not a lot is known about where these MPP cells are present. One hypothesis states these could be present in the pancreatic duct glandular structures (PDGs) that can be found as specialized compartments with a gland-like outpouching look (Figure 1) in the ductal epithelium (30, 35). The actual origin and location of the MPP that can evolve into islet cells is not known to date and thus needs to be further investigated for a better understanding of the potential origin of PNENs.
MOLECULAR (EPI)GENETICS
Genetic Syndromes
Although PNENs typically occur sporadically, approximately 10-20% of them develop in the context of hereditary syndromes. The syndrome most at risk for PNEN development is Multiple Endocrine Neoplasia (MEN1) (60%), an autosomal dominant disease caused by inactivating mutations in the MEN1 gene (10, 36-39). MEN1 is a tumor suppressor gene located on chromosome 11q13 that encodes for the nuclear protein menin which plays an important role in the PI3K/Akt/mTOR pathway, histone modifications, DNA repair mechanisms, and cell cycle control (10, 37, 38, 40, 41). In addition, 5 to 18% of the patients with von Hippel-Lindau (VHL) syndrome develop PNENs. These patients carry a germline mutation in the VHL gene located on the short arm of chromosome 3. The VHL protein can be found in different complexes that mediate ubiquitin-mediated degradation and stimulate angiogenesis (10, 42, 43). Other hereditary syndromes at risk include Tuberous Sclerosis (TS) and neurofibromatosis type 1, caused by mutations in TSC1, TSC2 and NF1, respectively (36-39).
Sporadic PNENS
Through next-generation sequencing of PNENs, it became apparent that there are distinct genetic differences, strongly depending on differentiation and functionality of the tumor (36).
For example, genetic analyses of F-PNENs revealed that insulinomas are often characterized by a hotspot mutation (p. T372R) in the Yin Yang 1 (YY1) gene in 30% of the Asian and 8-33% of the Western/Caucasian population (10, 36, 44). This recurrent mutation is located in the DNA binding domain of YY1, hence strongly affecting the DNA binding capacity of this transcriptional activator/repressor (44). In NF-PNENs, on the other hand, somatic mutations were most commonly identified in MEN1 (44.1%) followed by DAXX (25%) and ATRX (17.6%) (41). Atrx interacts with DNA methyltransferases (DNMT) 3A and 3L to form the Atrx-DNMT3A-DNMT3L (ADD) complex. This interaction is crucial for maintenance of histone methylation patterns in newly replicated chromatin, hence indirectly ensures correct gene expression. Moreover, Atrx also interacts directly with Daxx. In doing so, Daxx functions as a kind of chaperone for the deposition of histone variant H3.3 at the level of CpG islands, telomeric and ribosomal repeats and the rest of the genome. Consequently, a loss of ATRX and DAXX results in changes in DNA methylation patterns throughout the genome (45). In addition, mutations in PTEN, TSC2, and PIK3CA have already been reported in respectively 7.8%, 8.8% and 1.4% of PNENs, and they all affect the PI3K/Akt/mTOR pathway (41). Later, Scarpa and colleagues identified mutations in DNA repair genes MUTYH, CHEK2, and BRCA2 as well (39).
Distinct genetic differences could also be observed between G3 pancreatic NETs (PNETs) and pancreatic NECs (PNECs). The latter do not carry mutations in the known genes for PNETs (MEN1, DAXX, and ATRX), but instead appear to have mutations in TP53, RB1, KRAS, and CDKN2A/p16 (10, 36, 38). Considering that these mutations tend to result in altered protein expression, IHC might facilitate in distinguishing PNETs from PNECs, which have similar Ki-67 values. Nevertheless, results should always be interpreted with caution (10, 36, 46).
Besides point mutations, copy number alterations (CNAs) have also attracted attention. CNA patterns that were frequently identified included whole or partial loss of chromosomes 1, 2, 3, 6, 8, 10, 11, 15, 16, 21 and 22, while gains have been observed in chromosomes 5, 7, 12,14 and 17 (10). Moreover, PNENs appear to display very specific CNA patterns that allow to distinguish PNENs from the more common PAADs. Boons and colleagues therefore developed a classification model, based on tumor tissue, which demonstrated a sensitivity, specificity and area under the curve (AUC) of 100%, 95% and 100% in the validation cohort (47). Benign insulinomas tend to display lower rates of CNAs (36).
Since genes such as MEN1, DAXX and ATRX are of importance in several epigenetic regulatory processes, it was extremely likely that also epigenetic alterations commonly occur in PNENs. In fact, both in hereditary and sporadic PNENs promotor hypermethylation is observed in tumor suppressor genes, which is associated with silencing of gene expression (10, 48). Chan and colleagues checked whether methylation profiles and expression were different in the A-D-M mutated group versus the A-D-M WT group. They observed that both groups clustered in two separate clusters and even revealed that gene expression of the A-D-M mutated group was respectively high and low in the ARX and PDX1 gene and the latter gene also displayed hypermethylation. This profile appeared to be quite similar to that of α cells in the pancreas (28). These results were confirmed by Neiman et al., who observed high methylation levels in the PDX1 promotor region in α cells, while β cells tend to have low methylation in this region (49). Based on this PDX1 gene methylation, Boons and colleagues performed unsupervised hierarchical clustering and could subsequently observe two subpopulations, A and B, which respectively contained the α and β cells. Of note, the majority of the mutated PNENs was found in group A confirming the findings of Chan et al. (28, 50). These results suggest that methylation profiling of the PDX1 gene could potentially help to divide PNENs into distinct clinically relevant groups that have different prognosis and risk of relapse (50). Recently, three subgroups (T1, T2 and T3) of PNENs have been identified, based on their methylation profile. Here, the T1 group consisted of the A-D-M WT tumors, while the T2 subgroup encompassed the A-D-M mutated tumors with recurrent chromosomal losses and methylation in the gene body of the MGMT gene. The last group, T3, displayed mutations in MEN1 and recurrent loss of chromosome 11. Tumors found in the latter group tend to have a better prognosis (51).
DIAGNOSIS AND MONITORING
The gold standard for diagnosing PNENs remains an immunohistochemical examination of the tumor tissue, but imaging and serum markers are also extremely important in the diagnostic process. The clinical presentation often determines the sequence of examinations. For example, patients with F-PNENs will usually undergo a biochemical blood analysis first based on their hormonal symptoms, whereas NF-PNENs are often detected by chance on imaging (25, 52, 53).
Immunohistochemistry
To correctly classify PNENs, tumor morphology and proliferation rates (Ki-67 and mitotic index) should be evaluated in tissue biopsies. These are usually obtained from surgical specimens, percutaneous core biopsies, or preoperative biopsies (52, 54, 55). The latter were mainly derived from endoscopic ultrasound (EUS) guided fine-needle aspirations (FNA) which, in recent years, have been increasingly replaced by fine-needle biopsies (FNB) as these enable histological tissue samples to be obtained, hence immunohistochemistry (IHC) to be performed (56, 57). This immunohistochemical examination is most often initiated by confirming the neuroendocrine differentiation by checking CgA and synaptophysin (SYP) expression (52, 54). Other markers such as neuron-specific enolase (NSE) and CD56 are less specific, hence less useful (58). Next, tumor morphology is assessed to determine whether the PNEN is well- or poorly-differentiated (Figure 2). In general, well-differentiated PNENs are characterized by uniform cells with a finely granular cytoplasm and round to oval nucleus which are arranged in a trabecular, glandular, or tubuloacinar pattern (54, 59). Moreover, typically all cells have a heterogeneous expression of CgA in their cytoplasm, whereas SYP stains more diffusely. Poorly-differentiated PNECs, on the other hand, consist of atypical neoplastic cells that often lack CgA and even SYP (59, 60). Ultimately, tumors are graded by proliferation rate that is influenced by two parameters, Ki-67 and mitotic count. The latter is usually reported as the number of mitoses per mm2 which in practice is often complicated by a limited tissue area. The mitotically active regions are then measured again via IHC to determine the Ki-67 index (Figure 3). It is therefore logical that the Ki-67 index is usually higher than the mitotic count since it considers the entire mitotic process and not just the number of mitoses. If both values assign a different grade to the same tumor, the highest grade, associated with the worst prognosis, is assumed (17, 23, 52, 54).

Figure 2.
Hematoxylin-eosin IHC staining of (A) well-differentiated PNET and (B) poorly-differentiated PNEC. This figure has been adapted from Archives of Pathology & Laboratory Medicine, Fang J. M. and Shi J., A clinicopathologic and molecular update of pancreatic neuroendocrine neoplasms with a focus on the new world health organization classification, 143 (11): 1317-1326. © 2019 (59).
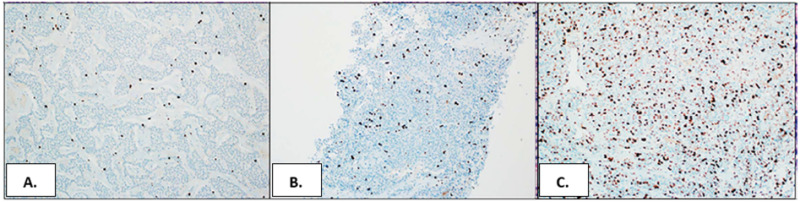
Figure 3.
(A) PNEN G1 with Ki-67 index of less than 3%. (B) PNEN G2 with Ki-67 index of 3% to 20%. (C) PNEN G3 with Ki-67 index higher than 20%. This figure has been adapted from Archives of Pathology & Laboratory Medicine, Fang J. M. and Shi J., A clinicopathologic and molecular update of pancreatic neuroendocrine neoplasms with a focus on the new world health organization classification, 143 (11): 1317-1326. © 2019 (59).
Imaging
Regardless of whether a PNEN is functional or non-functional, imaging is critical to assess the extent of the disease by localizing the primary tumor and identifying the size of metastatic disease. Localization is required preoperatively to increase the accuracy of intraoperative techniques and to reduce the need for repeated surgery. Besides, imaging is involved in patient’s management as it allows to monitor tumor growth and evaluate response to treatment (2, 25, 53, 55, 61). A multimodal approach is applied to diagnose and stage PNENs which comprises both anatomical and functional imaging modalities (2, 61-66).
ANATOMIC IMAGING
Anatomical imaging modalities such as Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) are capable of depicting normal and diseased tissue at high spatial resolution (67). Contrast enhanced CT is the most commonly used and preferred modality as it is widely available, renders clear anatomical images of the pancreas, lymph nodes, and liver metastasis and allows to assess vascular invasion and resectability (25, 52, 62, 64, 68, 69). The more recent, multiphase multidetector CT scan exhibits even more advantages including reduced artifacts due to rapid scan time, improved arterial phase images due to accurate contrast medium tracking and improved resolution by generating thinner slices that can be studied in different anatomical planes (61, 65). In addition, the frequent hypervascular nature of PNENs results in typical high contrast uptake in the arterial phase on CT which can aid in differentiating from pancreatic adenocarcinoma. The average sensitivity of contrast-enhanced CT varies from 63% to 83% and detection rates range from 69% to 94.3% (52, 67, 70). It appears that imaging of PNENs is often influenced by their biological heterogeneity. For example, gastrinoma become more apparent on postcontrast images and large NF-PNENs often have a necrotic or cystic appearance which tend to complicate diagnosis with imaging alone. In the latter case, MRI can be useful as cystic neoplasms can be better visualized due to the higher resolution, rendering MRI complementary to CT. MRI displays a similar sensitivity to CT (79%), but has some advantages over CT as it displays a good sensitivity even without administration of contrast agents, employs non-ionizing radiation, and is hence safer for patient follow-up (52, 53, 67). Limitations on the other hand include a higher frequency of motion-related artifacts as well as a longer acquisition time compared to CT (71).
Still, both conventional imaging modalities depend to a large extent on the tumor size (2, 25, 61, 72, 73). More than 70% of PNENs larger than 3 cm are detected, but only 50% of PNENs smaller than 1 cm are identified. As a result, small primary PNENs, especially insulinoma and duodenal gastrinoma, are frequently missed as well as small liver metastases (2, 25, 61, 62, 72-74). For small PNENs, which cannot be detected using CT and MRI, EUS is considered the predominant imaging technique (68). Because of the high spatial resolution of this modality, it is possible to localize even very small lesions (2-3 mm) (3, 74). Additionally, it is feasible to obtain high yield tissue samples by means of an FNA/B that can be used for Ki-67 measurements. Such EUS-FNA/B have a diagnostic accuracy of 80% for pancreatic adenocarcinoma and 46% for PNENs. In patients with proven insulinoma, EUS displays a sensitivity of 94% as a first-line modality. This renders EUS extremely valuable for localizing primary insulinoma (2, 68, 74, 75). However, EUS is not generally available, can be technically challenging, and results are operator dependent. In the hands of an expert, sensitivities of 79 to 100% can be achieved (61).
FUNCTIONAL IMAGING
Prior to the development of the current functional imaging modalities, selective angiography and sampling for hormone gradients were employed. However, due to the highly invasive nature of these techniques, minimally invasive modalities were developed which had a great impact on patient management (2, 53, 64).
Although PNENs exhibit highly heterogeneous biological behavior, 80-100% of PNENs, with the exception of insulinomas (50-70%), overexpress the G-linked protein somatostatin receptors (SSTRs), mainly subtypes SSTR-2 and -5. These receptors interact with somatostatin, a peptide hormone that affects neurotransmission and cell proliferation, but also the secretion of various compounds in the digestive system (2, 52, 62-64, 67, 76). Interestingly, these SSTRs also bind synthetic, radiolabeled somatostatin analogs (SSAs) with high affinity, which constitutes the basis of the primary functional imaging tool for PNENs, namely Somatostatin Receptor Imaging (SRI). SRI will not only allow to stage PNENs, but will also predictively identify patients eligible for SSA therapy (2, 52, 53, 64, 67, 68). One of the first SSAs used to target the SSTRs was octreotide labeled with 111Indium via chelator, diethylene-triamine-pentaaceticacid (DTPA). This 111In-DTPA-octreotide emits gamma rays that are detected 24 hours after intravenous injection using Single Photon Emission CT (SPECT) or SPECT/CT, the so-called Octreoscan® (53, 62, 64, 70, 77). The Octreoscan® is often combined with CT to improve the anatomic localization making it highly sensitive (77%), detecting 50-70% of primary PNENs, but less of the insulinomas and duodenal gastrinomas (2, 62-64, 68, 78-81). Major drawbacks include the availability and price of 111In-DTPA octreotide, the staggering acquisition time as well as the intrinsic shortcomings of SPECT, such as low spatial resolution (8-12 mm) (67, 82).
Positron Emission Tomography (PET) could provide better spatial resolution and greater precision (71). One of the most widely employed PET radiotracers currently used to image tumors is 18Fluor-labeled deoxy-glucose (18FDG). For high-grade NETs, especially NECs, 18FDG-PET/CT is a better choice as nuclear medicine modality as SSTR expression decreases when proliferation rates increase. 18FDG-PET can even be positive in G2 and G3 NETs. To this date, no cut-off value has been determined. However, it seems like neoplasms with Ki-67 values > 15% are more likely to exhibit a positive 18FDG-PET/CT, which is also a predictor of a more aggressive course (53, 81, 83, 84). However, 18FDG-PET/CT appears to be less useful in the majority of PNENs as these often show limited glucose uptake due to a rather slow growth rate (52, 64).
The development of new PET/CT radiotracers has been a major breakthrough in PNEN imaging. 11Carbon-5-hydroxytryptophan-labeled or 68Gallium-labeled SSAs including DOTA-tyrosine-3-octreotide (DOTA-TOC), DOTA-octreotate (DOTA-TATE), and DOTA-1-NaI-octreotide (DOTA-NOC) showed better sensitivity and diagnostic accuracy than the conventional imaging studies (Figure 4) and the Octreoscan® (Figure 5) (2, 52, 64, 71, 85-87). A meta-analysis revealed that 68Ga-DOTA-SSA PET for the diagnosis of NETs has a pooled sensitivity and specificity of 93% and 91%, respectively (88). Admittedly, the majority of the studies involved heterogenous populations, but most included a sizable minority of 20-30% PNENs. Hence, the overall data, although far from perfect, support use of 68Ga-DOTA-SSA PET over the Octreoscan® (89). Moreover, it is highly sensitive for the detection of bony metastases and it might obviate the need for additional radiologic studies. In addition to a higher sensitivity, other advantages of 68Ga-DOTA-SSA PET include patient convenience (imaging sessions take 70-90 minutes instead of 24 hours), lower radiation exposure, utility in finding unknown primary PNENs and it can lead to changes in treatment plans in about 33-41% of the patients (67, 89-93). 68Ga-DOTA-SSA PET might also be better at quantifying SSTR expression which facilitates targeted therapy such as PRRT (89). Consequently, 68Ga-DOTA-SSA PET quickly became the imaging modality of choice (67, 68, 71). However, similar to other imaging studies, false positives may occur due to pancreatic uncinate process activity, inflammation, osteoblastic activity, and splenosis (94). No doubt other PET agents will follow since PNENs express a variety of receptors for which there are potential ligands. For example, insulinomas express SSTRs in 50% of the cases, so tracers targeting the glucagon-like peptide-1 (GLP-1) receptor might be more useful in those patients (80, 95, 96).

Figure 4.
Overview of (A) 68Ga-PET/CT, multiphase (B) atrial and (C) portal vein CT scan images from patient with partially cystic PNEN. The arrow indicates a liver metastasis which is only visible on the 68Ga-PET/CT scan. This figure has been adapted from Current treatment options in oncology, Morse B., Al-Toubah T. and Montilla-Soler J., Anatomic and functional imaging of neuroendocrine tumors, 21 (9): 75 © 2020 (67).

Figure 5.
Comparison of (A) planar Octreoscan®, (B) Octreoscan®/SPECT/CT fusion, (C) planar 68Ga-DOTATOC-PET and (D) 68Ga-DOTATOC-PET/CT in the same patient. Images C and D clearly display a more precise delineation of the lesions. This figure has been adapted from International journal of endocrine oncology, Maxwell J. E. and Howe J. R., Imaging in neuroendocrine tumors: and update for the clinician, 2 (2): 159-68 © 2015 (82).
All benefits taken into account, the FDA approved 68Ga-DOTATOC PET in 2016 in the US, after being available in Europe for a number of years. Furthermore, with the development of an FDA approved 68Ga generator, an on-site cyclotron is no longer required, making this technology more widely available. A multi-society workgroup has recommended that 68Ga-DOTA-SSA PET replace use of Octreoscan®, unless it is not accessible, in combination with at least one anatomic imaging technique (66, 70).
Assessment Through Circulating Biomarkers
As stated earlier, the current gold standard for diagnosing and molecularly profiling PNENs remains the analysis of surgical or biopsy tissue samples. However, these samples have a highly invasive character, rendering repetitive sampling unfeasible. Further limitations are the individual patients’ risk and procedural costs. Besides, they represent merely a snapshot of tumor heterogeneity, which strongly influences accuracy. Hence, liquid biopsies aroused strong interest since they form a cost-effective and minimally invasive way to analyze the tumor’s behavior. The most frequently used source is blood as it allows to examine the so-called tumor circulome that consists of a set of circulating components that originate from the tumor (97-101). These blood-based biomarkers play a pivotal role in diagnosing and staging PNENs, monitoring response to therapy, and detecting tumor progression. In case of F-PNENs, specific circulating biomarkers such as insulin, gastrin, and glucagon are employed in hormonal assays to correctly diagnose F-PNENs. Moreover, both F- and NF-PNENs frequently secrete non-specific markers including CgA, neuron-specific enolase (NSE), pancreastatin, etc., which can be detected in patients’ blood as well (2, 9, 13, 52, 53, 55, 102). Besides circulating proteins, PNENs also shed circulating tumor cells (CTCs), circulating tumor RNA (ctRNA) and DNA (ctDNA) which could serve as potential biomarkers (98-100, 103, 104).
SPECIFIC BIOMARKERS AND HORMONAL ASSAYS
Depending on the type of F-PNENs (outlined in Table 1), specific biochemical tests are performed. When insulinoma is suspected, serum levels of insulin and C-peptide are measured at a confirmed hypoglycemia during prolonged period of fasting (approximately 72 hours) as patients present with increased levels (> 6 µU/L and 0.6 ng/mL, respectively) even when glucose levels are low (7, 31, 52, 53, 104). In case of gastrinomas, the serum gastrin levels will be 10 times higher than the upper limit and gastric pH will be lower or equal to 2 (3, 7, 52, 53, 105). In patients with suspected VIPoma and glucagonoma, diagnosis is confirmed by determining the fasting levels of VIP and glucagon (7, 53).
Chromogranin A (CgA)
CgA, a glycoprotein stored in and secreted by the secretory granules of the neuroendocrine cells, plays an important role not only in immunohistochemistry, but also as a circulating marker (7, 17, 52, 53, 102). CgA is useful as a marker for both functional and non-functional PNENs, as elevated levels are noted in 50-100% of the patients with PNENs (2, 106-108), depending upon the histological subtype (104, 109, 110). For example in gastrinoma, CgA levels are consistently high due to gastrin-induced enterochromaffin-like cell hyperplasia (111), while insulinoma show significantly lower levels of circulating CgA (112). Besides, blood levels depend upon malignant nature of the tumor, tumor burden, and progression, hence small tumors may be associated with normal CgA levels (113, 114).
The ENETS still recommends the use of circulating serum CgA as marker during diagnosis and follow-up in NF-PNENs (7, 115). However, the actual diagnostic value of this marker is still questionable (115). Sensitivity, specificity and overall accuracy of this clinical biomarker equal 66%, 95% and 71%, respectively (7), but these values tend to vary according to the specific assays and diagnostic threshold (52). Common conditions that can falsely elevate CgA levels, thus impair specificity, include decreased renal function, treatment with proton pump inhibitors (116), and even essential hypertension (117). In addition, 30-50% of NENs do not show elevated CgA levels, limiting sensitivity (47, 115, 118). This group mostly involves small, localized, non-functional NETs where CgA levels are normal in approximately 70% of the cases (119). As a result, these patients are in a higher need for accurate biochemical markers as diagnosis is harder both clinically and by use of imaging techniques. Moreover, SSA treatment cause a decrease in CgA secretion, which is why results should always be interpret with caution (104). In terms of follow-up, prospective studies demonstrated that elevated CgA levels do not correlate with imaging and tumor progression, hence questioning the potential of CgA as follow-up biomarker (115). Compared to CgA, CgB is not impacted by for example proton pump inhibitor treatment (112, 116). However, only in 25% of the cases with elevated CgA levels, CgB was elevated as well, thus routine estimation of CgB in all patients seemed not informatic in clinical practice (120).
Neuron-Specific Enolase (NSE)
NSE is a glycolytic enzyme expressed in the neuroendocrine cells of which levels can be elevated in PNEN patients, particularly those with a poorly-differentiated tumor (58, 110). However, its clinical use is limited as a blood-based biomarkers for NETs because sensitivity and specificity are only 39-43% and 65-73% to distinguish NETs from non-NETs. Consequently, NSE is therefore inferior to CgA in clinical practice (17, 110, 121). When combined with CgA measurement, sensitivity improves and reliability of NET diagnoses increases. Moreover, elevated CgA/NSE levels appear to provide prognostic information on progression-free survival (PFS) and OS (7, 102, 122).
Pancreastatin
Pancreastatin, a post-translational processing product of CgA, is suggested to be a useful prognostic marker of NETs as pre-treatment levels > 500 pmol/L are an independent indicator of poor prognosis. Moreover, this marker is reported to correlate with the number of liver metastases and an increase in pancreastatin levels after treatment with SSAs is associated with poorer survival (52, 123). For diagnosis of NETs, pancreastatin is less sensitive than CgA, but also less susceptible to non-specific elevation (52, 121).
Pancreatic Polypeptide (PP)
PP is a hormone predominantly produced in pancreatic polypeptide cells, located in the head of the pancreas (7, 17, 109, 124). When used alone, a sensitivity of 63% is achieved in PNENs, but when combined with CgA sensitivity increases to 94%, better than either marker alone (109). However, less than 50% of PNEN patients display elevated serum levels and increases do not correlate with tumor burden and/or aggressiveness (102, 124). Moreover, there are several clinical conditions that can induce falsely elevated levels such as physical exercise, hypoglycemia, and food intake, whereas diarrhea, laxative abuse, high age, inflammatory processes and chronic renal disease could lead to a decrease (7, 102).
Other Protein-Based Markers
Besides the above-mentioned markers, ProGRP and Neurokinin A can be used to further improve diagnostic and especially prognostic information. ProGRP in fact stimulates cell proliferation which is why increased levels are often associated with a more aggressive tumor and therefore worse prognosis (7, 12). In addition, several markers were reported to be useful for the detection of bone metastases that can be either osteolytic or osteoblastic. Bone alkaline phosphatase (BAP) indicates osteoblast function, while urinary N-telopeptide reflects osteoclast activity or bone resorption. An increased osteoclast activity predicts a poor outcome (12, 125, 126).
Circulating Tumor Cells (CTCs)
CTCs have been investigated in a wide range of tumor types, and have gained increased interest in PNENs due to the limitations of the current circulating markers (98-100, 104, 121, 127, 128). The recently developed platform, CellSearch®, allows to detect and isolate CTCs based on expression of the epithelial cell adhesion molecule (EpCAM) on the cell membrane. EpCAM is a transmembrane epithelial glycoprotein that is overexpressed in adenocarcinoma, but recent studies (127-129) revealed EpCAM positivity in ileal, pancreatic, unknown primary, and gastric NETs as well. However, only 21-24% of the metastatic PNEN patients had detectable CTCs in the blood stream, which could potentially be explained by a slow shedding of CTCs or loss of EpCAM expression. Presence of CTCs was associated with increasing tumor burden and grade, while CgA failed to reveal this relationship. Changes in CTC levels were associated with treatment response and OS, revealing its potential as marker during treatment follow-up (127, 129). Furthermore, presence of CTCs could distinguish between patients suffering from PNENs with and without bone metastases with an area under the curve (AUC) of 79% (130). A phase II PAZONET study, during which Pazopanib treatment was evaluated, even demonstrated that patients without baseline CTCs showed improved response and longer median PFS (131). Contrarily, the CALM-NET phase IV study reported no notable effect of the presence of CTCs at baseline on PFS in patients treated with Lanreotide (132). Lastly, CTCs provide the opportunity to detect (epi)genetic alterations in PNENs through DNA and RNA extraction, but they can also be used to determine the SSTR status via immunohistochemistry which could facilitate therapeutic management (133, 134).
Circulating Tumor DNA (ctDNA)
ctDNA is the fraction of cell-free DNA (cfDNA) that originates from the tumor and constitutes one of the most promising new markers. It provides a representation of the whole tumor and contains tumor-specific genetic and epigenetic alterations, which allow to distinguish healthy from tumoral DNA (98-100, 104, 135). However, ctDNA research regarding NENs is still in its infancy. Boons et al., published the first paper confirming the presence of ctDNA in the plasma of metastatic PNEN patients by looking for tumor-specific single nucleotide variants (SNVs) via custom digital droplet PCR (ddPCR). In patients with localized PNENs, ctDNA could not be detected (136). In the same study, they revealed a significant correlation between CNA profiles of PNEN tissue and ctDNA and demonstrated the feasibility to detect ctDNA using these profiles (136). These findings were exploited in a more recent study, where they performed a cfDNA CNA analysis in a cohort of 43 NEN patients. Using this analysis, ctDNA could be detected in 13 of the 21 PNEN patients. ctDNA positivity appeared to be significantly associated with higher WHO grade, location of the primary tumor and higher levels of CgA and NSE. Besides, a worse OS was observed in ctDNA-positive patients. In addition, they illustrated that CNA patterns in cfDNA could even assist in distinguishing PNENs from the more common PAADs. Moreover, the longitudinal tumor fraction (i.e., amount of ctDNA vs. total cfDNA) measurements were associated with PFS and could indicate tumor progression (47).
MicroRNA (miRNAs)
miRNAs are short noncoding RNAs (< 30 nucleotides) designated to regulate many processes including cell proliferation, apoptosis, and development (134, 137), by inducing translational repression or degradation of certain mRNAs (138). In cancer, miRNA regulation is often altered as is the case in PNENs (138). Normal pancreatic islets and PNENs display a distinctly different miRNA profile as PNENs express miRNA-103 and-107, but lack miRNA-155. A set of 10 miRNAs was even able to perfectly distinguish 40 PNENs from 4 PAADs (137). miRNA-204 was overexpressed in insulinomas only, miRNA-196a had a prognostic function and overexpression of miRNA-21 was associated with higher Ki-67 rates and presence of liver metastasis (137). A more recent study, demonstrated that the combination of a set of miRNAs together with CgA measurements could improve diagnostic accuracy (139). However, data on circulating miRNA is still scarce as miRNA measurements in NETs are not properly standardized, requiring further research (140).
NETest
The NETest, a blood-based multi-analyte transcript assay, was developed in 2013 by Modlin and colleagues (141). The expression of 51 marker genes, encompassing genes associated with NENs, is examined using a quantitative PCR (qPCR) and analyzed using multivariate algorithms (142, 143). These algorithms enable the calculation of a disease-activity score, ranging from 0 to 100, with scores higher than 20 representing tumoral samples (140, 142, 143). The NETest captures accurate diagnosis and tumor biology of NETs with the most recent study demonstrating an accuracy of more than 91%. More specifically, the NETest has proven useful for diagnosing PNEN patients, as PNENs could be distinguished from other pancreatic malignancies with an accuracy of 94% (142, 143). The test also shows to be a real-time monitor of clinical status through the disease-activity score of NEN patients. Low biological activity corresponds to a score of less than 40, while intermediate and high biological activity, indicating tumor progression, have scores of 41-79 and 80-100, respectively (103, 140, 143). Stable and progressive disease could be differentiated with an accuracy of 84.5-85.6%, consistent with image-based categorizations (103). Moreover, changes in NETest disease-activity scores over time correlated with response to treatments including SSAs, PRRT, and surgery (144-147). For example, in a prospective analysis, performed by Modlin and colleagues, 35 pancreatic and small intestine NEN patients were included that all displayed elevated NETest levels prior to surgery, while only 14 of them had increased CgA levels. After tumor removal, the disease-activity scores reduced from 80 ± 5 to 29 ± 5 (p < 0.0001), whereas changes in CgA levels did not correlate with resection. Four of the 11 patients with complete tumor resection still presented increased NETest scores one month after surgery and showed positive evidence of recurrence 6 months post-surgery (144).
Since 2013, the NETest has proven to perform better than the single analyte tests (e.g., CgA) and these results appeared to be highly robust and reproducible (103, 140, 142). However, a large independent validation study conducted in the Netherlands has revealed that the test is more sensitive, but less specific than CgA suggesting its suitability as a marker for disease follow-up, but not as a screening tool (147). This test is not affected by food intake or specific medication, is easy to use and available which all increases clinical utility (140). The NETest possess advantages from an economic point of view too. Identifying patients with molecularly stable disease (SD) could potentially lead to fewer use of imaging modalities. Moreover, by enabling faster identification of the clinical status than with imaging, ineffective therapies can be ceased more quickly with another obvious cost-benefit effect (140, 142). Despite all advantages, NETest is currently not implemented in a clinical setting. Results of additional independent validation studies and other practical aspects such as costs and transparency will ultimately determine its integration in clinical practice.
MANAGEMENT OF PNENS
With a better understanding of the heterogeneity in PNENs, the number of treatment options has increased substantially over the years. Unfortunately, there is a lack of head-to-head comparison data. Therefore, treatment must be individualized considering the age and overall health of the patient, the specific toxicities of potential treatment(s), costs, and potential impact on quality of life (QOL). Consequently, decisions with regard to patient management must be made by an experienced, multidisciplinary team together with the primary care physician (52). Generally, the management of PNEN patients consists of a series of well-defined steps. These comprise of: 1) establishing a diagnosis, 2) determining localization and extent of tumor, 3) controlling hormone excess state in case of F-PNENs, 4) resecting tumor, if possible, 5) checking for presence of hereditary disease (MEN1), 6) treating advanced and metastatic PNENs, and 7) long-term monitoring for tumor progression (63, 148).

Figure 6.
It is crucial to consider grade/differentiation, stage/extent, and functional status of the tumor as different treatment schemes evolved based on these factors (Figure 6). For example, surgery is usually advocated for PNENs that are functional, larger than 2 cm, or intermediate-to-high grade (3, 8, 52). For patients with metastatic disease, the treatment options are extensive and encompass surgical debulking, systemic therapies including chemotherapy, or targeted therapy such as liver-directed therapy and peptide receptor radionucleotide therapy (PRRT) (149). It is not unusual for the management plan to change based on treatment response and disease progression. Failure to respond to treatment or unexpected changes in the tempo of disease due to tumor dedifferentiation and tumor heterogeneity are well-described in PNENs. Accordingly, most patients will receive multiple treatments during the course of their disease, but there is no data on the optimal treatment sequence (52, 105). The various treatment modalities are discussed below.
Surgical Management
Surgery continues to play a major role in the management of patients with PNENs as it remains the only potentially curative treatment for PNEN patients and it can alleviate clinical symptoms caused by excessive hormone production and tumor bulk (2, 3, 7, 149-151). Furthermore, several studies revealed that patients who underwent surgical resection had a reduced risk of metastases as well as showed an improved disease-free survival (DFS) (152, 153). Different approaches exist such as resection of the primary tumor and surrounding lymph node metastases through pancreaticoduodenectomy (Whipple procedure) and pancreatectomy (central or distal) as well as the more conservative methods including sparing enucleation and wait-and-see observations (7, 52).
Choosing the appropriate approach depends on the extent and location of the tumor, the functional status as well as the presence or absence of metastases (52, 150, 154-156). Generally, surgery is recommended in patients with localized NF-PNENs. Besides the primary tumor, peritumoral metastases should be eradicated as well since nodal metastases occur in at least 30% of NF-PNEN patients which affect tumor grade, but more importantly DFS (157, 158). Exceptions occur in patients with sporadic, low-grade (G1/G2) NF-PNENs smaller than 2 cm (3, 8, 52, 149, 150, 154-156). For those patients, optimal management is controversial as some recommend surgical interventions such as enucleation, whereas others including the ENETS advocate a wait-and-see attitude due to the indolent nature of these tumors (3, 8, 52, 148-150, 159, 160). A similar conservative approach is encouraged in MEN1 patients with NF-PNENs of 2 cm or smaller as these tend to have a low disease-specific mortality (161). Thakker and colleagues, on the other hand, suggest resection of NF-PNENs larger than 1 cm that demonstrate significant growth over 6 to 12 months (162).
Surgical excision of the tumor is also recommended for patients with F-PNENs as these display high cure rates (2, 13, 149, 163-165). The National Comprehensive Cancer Network (NCCN) guidelines describe that insulinomas and gastrinomas are preferably removed by enucleation with peritumoral node dissection if the tumor is located in the head of the pancreas. Deeper, more invasive tumors are more appropriately eradicated by pancreaticoduodenectomy. The former strategy should also be applied for small peripheral glucagonomas and VIPomas. PNENs in the distal part of the pancreas, in turn, are ideally removed through distal pancreatectomy (149). Surgery for MEN1 patients with NF-PNENs and gastrinoma remains controversial as they often present with multiple primary tumors which renders curative surgery almost impossible. Aggressive resection of all PNENs smaller than 2 cm in MEN1 patients seems contra-indicated as several studies revealed that these patients rarely develop advanced disease and have a good prognosis (2, 18, 75, 165-167).
The traditional surgical approach is open laparotomy as this allows thorough abdominal exploration including bimanual palpation and intraoperative ultrasound of the pancreas and liver (2, 168). However, several studies reported that certain lesions in particular those amenable to enucleation or to distal pancreatectomy may be approached with laparoscopic or robotic techniques (169). Venkat and colleagues even demonstrated that patients who underwent laparoscopic resection had less blood loss and a lower overall complication rate, and were consequently permitted to leave the hospital sooner than patients who had had open pancreatic resection (170). Gastrinomas form an exception since palpation plays an important role in the detection of these often small malignancies. Moreover, 60-90% of these patients will have lymph node metastases in addition to the primary tumor (169, 171). Adopting a purely laparoscopic approach to these tumors will depend upon improvements in haptic feedback technology. For tumors requiring a Whipple procedure both laparotomic and laparoscopic approaches are used in centers worldwide as the latter is still associated with technical difficulties. However, when performed by trained hepatobiliary or laparoscopic surgeons’ effectiveness and safety are similar and, in some cases, even superior to open surgery (168, 171).
In patients with distant metastases, surgical intervention remains important, although it may no longer result in cure (52). The most common site of distant metastasis is the liver since 46-93% of NET patients develop liver metastases which can lead to liver failure, a common cause of death (52, 172-174). There are multiple options available for patients with hepatic metastases, including surgical resection which, in selected cases, appears to improve survival in uncontrolled series (157). The optimal approach depends on several factors including the extent of primary tumor and liver metastases, planned treatment as well as the age and overall health of the patient. Accordingly, the NCCN recommends complete resection (R0 resection) of primary tumor and liver metastases, if possible and otherwise consider tumor debulking (149). Aggressive resection of the primary tumor in the setting of liver metastases is associated with a survival benefit as both obstruction and further metastatic spread may be prevented. The 5-year survival rate after this surgery ranged from 65% to 73% which is significantly better than that of patients with nonresectable metastases (20%) although this difference might be at least partially explained by selection bias, where only very fit patients receive surgery (174-176). In case of the latter, numerous non-surgical options are available (see liver-directed therapy) and primary tumor, when found to be asymptomatic and stable, is not removed (52, 177). However, R0 resection can only be achieved in 10-20% of the cases as the majority of patients presents with multifocal and bilateral metastases and studies suggest that only one third of all liver metastasis are visible on imaging (52, 174, 175, 178, 179). Consequently, cytoreductive hepatic surgery is more frequently opted for, but this approach remains controversial as it is incomplete and the target population is not clearly described. It is therefore generally considered that patients with metastatic G1/G2 PNEN in which preferably less than 25% of the liver is affected are eligible for tumor debulking (2, 52). Several studies already showed that this procedure can alleviate clinical symptoms in F-PNENs, but also provide better long-term survival (2, 52, 149, 180-183). Moreover, debulking may also be associated with an improved response to concomitant therapy such as embolization (184). Radiofrequency ablation (RFA) is increasingly used in PNEN patients to address hepatic metastases and is often performed during surgery or laparoscopically (2). Patients with extensive liver involvement, who are consequently ineligible for R0 resection and tumor debulking, may be aided with a liver transplant to improve life expectancy (52, 176, 185). A non-randomized study in 88 patients who met strict criteria of transplant eligibility reported a difference in OS in the transplant (88.8%) and no transplant group (22.4%) after 10 years (185). Important concerns are the availability of the grafts as well as the lifelong immunosuppression required after transplantation. Also, the exact criteria for eligibility are very similar to those for tumor debulking which makes patient selection difficult (52).
Medical Therapy
Use of medical therapy is limited to those with locally advanced or metastatic disease. Some of the current and promising options for targeted systemic therapy are shown in Figure 7.
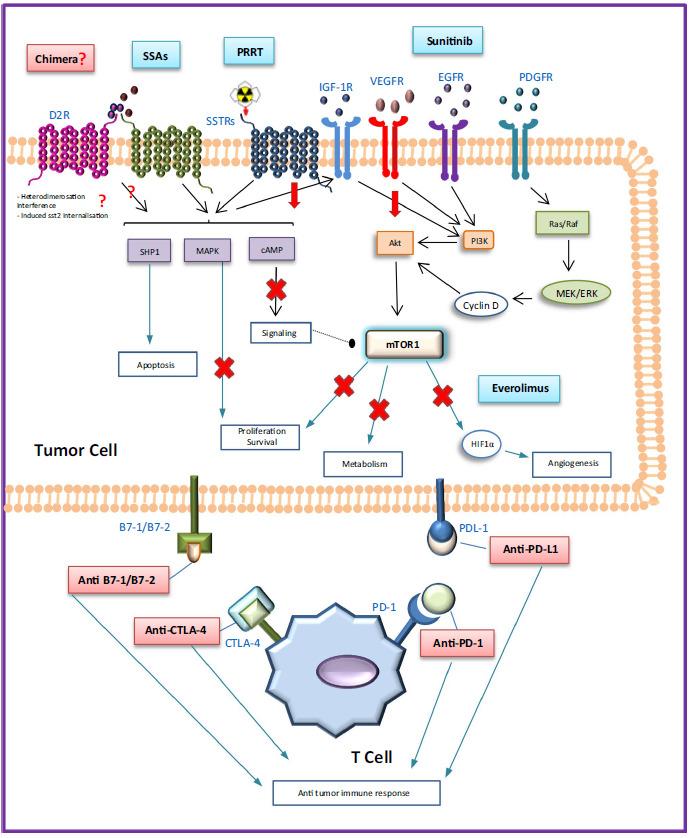
Figure 7.
Overview of the current (blue) and promising (red) options for targeted medical therapy in (P)NETs. This figure has been retrieved from Drugs, Herrera-Martinez A. D., Hofland J., Hofland L. J., Targeted Systemic Treatment of Neuroendocrine Tumors: Current Options and Future Perspectives 79:21–42 © 2019 (186).

Figure 8.
Visualization binding affinity of each of the three FDA-approved SSAs to the different SSTR subtypes. This figure was retrieved and adapted from Drugs, Herrera-Martinez A. D., Hofland J., Hofland L. J., et al., Targeted systemic treatment of neuroendocrine tumors: current options and future perspectives, 79:21-42. © 2019 (186).
SOMATOSTATIN ANALOGS
As previously described in the functional imaging section, SSTRs are often highly overexpressed in PNENs. Several SSAs including Octreotide (Sandostatin®) and Lanreotide (Somatuline®), which have affinity for different SSTR subtypes (Figure 8), were therefore marketed. These inhibit hormone secretion and thus reduce clinical symptoms in patients with F-PNENs (149, 186). Additionally, several studies revealed that SSAs are also capable to control tumor growth with a positive impact on PFS. The antiproliferative effect of SSAs in PNETs was confirmed in the Controlled Study of Lanreotide Antiproliferative Response in Neuroendocrine Tumors (CLARINET) trial. A total of 204 patients with well-differentiated, progressive NETs were included who were then randomly assigned to either Lanreotide or placebo treatment for 96 weeks. After 24 weeks, PFS rates were 65.1% and 33.0% in the Lanreotide and placebo groups respectively (Figure 9). The study also demonstrated that there was no significant difference in QOL and OS in both groups (149, 186-189).

Figure 9.
As an extension to the core CLARINET study, the CLARINET open-label extension (OLE) reported long-term safety and additional efficacy data. For this purpose, 88 patients with SD were selected from the core study. Forty-one patients continued their Lanreotide treatment, while 47 patients shifted from placebo to Lanreotide. Safety and tolerability were favorable during a mean treatment period of 40 months. In addition, adverse effects, that were either attributable or not to Lanreotide, were found to improve as duration of treatment, hence exposure, increased. Median PFS in patients who had already received Lanreotide in the core study was estimated at 32.8 months (Figure 10). Of the 32 placebo-treated patients who exhibited progressive disease (PD) in the core study, 17 patients persisted in PD, while the remaining 15 patients had a median time to progression (TTP) of 14 months (187, 190). Based on the findings from the CLARINET trial, the use of SSAs as first-line treatment for symptom relief and tumor control is recommended in the NCCN and ENETS guidelines for advanced, well-differentiated, unresectable PNENs, particularly those with a high burden of liver metastases (149, 188, 191).

Figure 10.
PFS of patients that received Lanreotide in the core and OLE CLARINET study. The OLE data is only visualized for patients that were originally assigned to and continued the Lanreotide treatment. This figure was retrieved and adapted from Endocrine-related cancer, Caplin M. E., Pavel M., Cwikla J. B., et al., Anti-tumor effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study, 23 (3): 191-9 © 2016 (190).
MOLECULAR-TARGETES AGENTS
Newly developed molecular-targeted treatments including Sunitinib and Everolimus (Figure 7) have shown to improve PFS in advanced, metastatic PNENs and represent the most common second line treatments that are currently available (52, 149). An overview of the most recent findings can be found in Table 3.
The tyrosine kinase inhibitor (TKI), Sunitinib, has been approved for the treatment of patients with well-differentiated, unresectable, locally advanced or metastatic PNENs as it displays an antiangiogenic working mechanism. It actually inhibits vascular endothelial growth factor receptors (VEGFR) 1 and 3, stem cell factor (SCF) receptor as well as platelet-derived growth factor receptors (149, 186, 192). A two-cohort phase II study, examining 107 patients with advanced NETs (of which 66 PNENs), reported an overall objective response rate (ORR) of 16.7% and SD in 68% of PNEN patients. Median TTP was 7.7 months in PNENs and 10.2 months in carcinoid patients (193). A phase III multinational, randomized, double-blind, placebo-controlled trial (SUN 1111) confirmed the activity of Sunitinib in patients with advanced, well-differentiated PNENs (Figure 11A). A total of 171 patients were enrolled in this study. Median PFS was 11.4 months in the Sunitinib group compared to 5.5 months in the placebo group, with the latter group having a higher mortality rate (25% vs 10%) (194). A retrospective analysis of the previous phase III trial reported an increased PFS in both the Sunitinib and placebo group (12.6 vs. 5.8 months). Median OS after 5 years were 38.6 and 29.1 months of the Sunitinib and placebo groups, respectively. Important to note here is that 69% of the placebo-treated patients shifted to Sunitinib treatment (195). Sunitinib presented with an acceptable safety profile as the most frequent adverse effects in the sunitinib group included diarrhea, nausea, vomiting, asthenia, and fatigue which can be managed through dose interruption or modification (192, 194, 196).
Everolimus (Afinitor®) is an oral, protein kinase inhibitor of the mammalian target of rapamycin (mTOR) pathway that displays proven antitumor activity in advanced PNENs, either alone or combined with Octreotide therapy. A multinational phase II study, the RADIANT 1 trial, has reported the efficacy of Everolimus alone and in combination with Octreotide in patients with metastatic PNENs that have progressed on chemotherapy (197). Monotherapy with Everolimus provided SD in 67.8% of patients and a partial response (PR) in 9.6%, while combination therapy resulted in 80% SD and 4.4% PR. Everolimus treatment also led to a decrease in CgA and NSE levels in 50.7% and 68.2% of the patients (Table 4). An early tumor marker response (i.e., > 50% decrease by 4 weeks) was associated with a significantly longer PFS (197). The RADIANT 3 trial, later on, investigated Everolimus as first line therapy in patients with advanced PNENs (Figure11B). Four hundred and ten patients with radiologic progression of disease were randomized to either Everolimus (10 mg once daily) or placebo. The median PFS was 11 months with Everolimus compared to 4.6 months with placebo representing a 65% reduction in estimated risk of progression or death. The proportion of patients alive and progression free at 18 months was 34% with Everolimus compared to 9% with placebo. Toxicities were mostly grade I or II (198). Similar PFS rates were reported regardless of whether patients were chemo-naïve or had received prior chemotherapy (199, 200). Addition of Pasireotide to Everolimus did not improve PFS compared to Everolimus alone (201).
Based on the recent, above-mentioned data, the European Society for Medical Oncology (EMSO) guidelines 2020 recommend the use of molecular-targeted agents such as Sunitinib and Everolimus in advanced, progressive PNENs (G1/G2) (191). Likewise, the North American Neuroendocrine Tumor Society (NANETS) guidelines 2020 recommend both treatments for well-differentiated, metastatic PNETs (G1/G2) (202). Both guidelines state there is no support to use Sunitinib nor Everolimus in treatment of PNET G3 or PNECs (191, 202). When comparing both molecular-targeted agents, response rates appear comparable (Figure 11). Since there has been no trial comparing the two agents directly, choice of agent may be based on the potential side-effects and patient’s overall health. For example, in patients with poorly-controlled hormonal symptoms, especially hyperinsulinism, congestive heart failure, hypertension, high risk of gastrointestinal bleeding or a history of myocardial infarction or stroke, Everolimus is thought to be the preferred choice (194, 202, 203). On the other hand, in patients with poorly controlled diabetes mellitus, pulmonary disease, or high risk of infection, sunitinib would be a more appropriate choice (192, 203). Moreover, up until now several biomarkers have been identified that correlated with the patient’s outcome. An overview of the currently known biomarkers can be found in Table 4 (204).
Table 3.
Results from Most Important Phase II and III Studies of Sunitinib and Everolimus in PNENs
| Study | Patients | Active treatment | PD at entry | ORR | PFS/TTP (months) | Safety and other comments |
|---|---|---|---|---|---|---|
| Sunitinib | ||||||
| Phase II, open label (193) | N = 107
| 50 mg daily Schedule 4/2* | No | ORR = 16.7% SD = 68% ORR = 2.4% SD = 83% | TTP = 7.7 TTP = 10.2 | G3 fatigue: 24.3% |
| Phase III, RCT, SUN 1111 (194) [Retrospect] (195) | N = 171
| 37.5 mg daily CDD** | Yes | ORR = 9.3% SD = 63% PD = 14% ORR = 0% SD = 60% PD = 27% | PFS = 11.4 [PFS = 12.6] PFS = 5.5 [PFS = 5.8] | Common AEs: 30%: diarrhea, nausea, asthenia, vomiting and fatigue 10-12%: G3/4 neutropenia and hypertension |
| Everolimus | ||||||
| Phase II, open label, RADIANT 1 (197) | N = 160
| 10 mg daily 10 mg daily + 30 mg LAR Octreotide | Yes | PR = 9.6% SD = 67.8% PD = 13.9% PR = 4.4% SD = 80% PD = 0% | PFS = 9.7 PFS = 16.7 | Specific AEs: 5.2%: G3/4 asthenia 8.9%: G3/4 thrombocytopenia Common AEs: 30%: stomatitis, rash, diarrhea, fatigue, nausea |
| Phase III, RCT, RADIANT 3 (198) | N = 410
| 10 mg daily | Yes | PR = 5% SD = 73% PR = 2% SD = 51% | PFS = 11 PFS = 4.6 | Common AEs: 64%: stomatitis 49%: rash 34%: diarrhea 31%: fatigue 23%: infections |
Abbreviations: ORR, objective response rate; PFS, progression-free survival; TTP, time to progression; SD, stable disease; PD, progressive disease; CDD, continuous daily dosing; AE, adverse event; LAR, long-acting release; PR, partial response; RCT, randomized clinical trial
- *
Concomitant use of SSA in 27% of PNET patients and 54% of patients with carcinoid tumors.
- **
Concomitant use of SSA in 26.7% of patients.

Figure 11.
This figure compares the PFS in patients with advanced metastatic PNENs, (A) treated with Sunitinib in the SUN 1111 trial (194) and (B) Everolimus in the RADIANT 3 trial (198). Figure A was retrieved and adapted from The New England journal of medicine, Raymond E., Dahan L., Raoul J. L., et al., Sunitinib malate for the treatment of pancreatic neuroendocrine tumors, 364 (6): 501-13 © 2011 (194). Figure B was retrieved and adapted from The New England journal of medicine, Yao J. C., Shah M. H., Ito T., et al., Everolimus for advanced pancreatic neuroendocrine tumors, 364 (6): 514-23 © 2012 (198).
Table 4.
Current Soluble Biomarkers and Correlations with Outcomes with Targeted Therapies in PNENs
| Study | Biomarker | Results |
|---|---|---|
| Sunitinib (204, 205) | VEGF | Increased in 53% of patients after 4 weeks of treatment Return to baseline after 2 weeks off treatment When Sunitinib level > 50 ng/dL higher changes observed No significant difference between PNET and carcinoids |
| sVEGFR | Decrease of ≥ 30% in sVEGFR-2 and -3 levels Return to baseline after 2 weeks off treatment Reduction in sVEGFR-3 correlated with better OR and PFS Lower baseline sVEGFR-2 with radiological SD for > 6 months Elevated baseline sVEGFR-2 correlated with improved OS | |
| IL-8 | Increase (>2-fold) in 43% and (>3-fold) in 23% of patients after 4 weeks of treatment Return to baseline after 2 weeks off treatment Increase (1.8-fold) after 4 weeks on treatment | |
| SDF-1α | Increase (20%) after 4 weeks on treatment Elevated baseline correlated with significantly shorter TTP, PFS and OS | |
| Everolimus (197, 206) | CgA | Increase (> 2-fold) of CgA at baseline correlated with decreased PFS and OS Reduction of > 30% in CgA levels after 4 weeks correlated with increased PFS and OS |
| NSE | Elevated NSE levels at baseline correlated with decreased PFS and OS Reduction of > 30% in NSE levels after 4 weeks correlated with improved PFS |
Abbreviations: VEGF, vascular endothelial growth factor; sVEGFR, soluble VEGFR; SDF-1α, stromal cell-derived factor 1 alpha. Note: This table was adapted from Molecular Diagnosis and Therapy, Mateo, J., Heymach, J. V. and Zurita, A. J., Biomarkers of response to Sunitinib in gastroenteropancreatic neuroendocrine tumors: current data and clinical outlook, 151-161. © 2012 (204).
CYTOTOXIC CHEMOTHERAPY
There is currently no unanimity on which cytotoxic chemotherapy would be optimal for the treatment of PNENs. Therefore, patient selection is key so factors such as primary tumor site and stage, differentiation, and proliferation index should be considered. Conventional cytotoxic chemotherapy is often used as first-line therapy for metastatic and progressive PNETs or PNECs (149, 207). ENETS guidelines describe the following indications: progression under SSA treatment, worsening clinical symptoms, and/or Ki-67 values > 10% (208). In a neoadjuvant setting, chemotherapy can play a potential role in tumor shrinkage prior to resection (7). Two major types of chemotherapeutic agents can be distinguished namely alkylating and platinum agents (7, 149, 207). In practice these are often combined with antimetabolites and anthracyclines (209). An overview of the most commonly used combinatory therapies and when to employ them is described in more detail below.
Alkylating agents such as Streptozocin, Dacarbazine, and Temozolomide are key in the treatment strategy of PNEN patients since they are often employed as second line treatment after progression under SSA (207). First of all, Streptozocin, a nitrosourea alkylating agent, is taken up by cells via a glucose protein 2 (GLUT2) after which cell damage is induced. Since the compound is associated with significant renal and hematological toxicity in high doses, it is often combined with 5-fluorouracil (5-FU) or Doxorubicin with dose reduction, hence reduced toxicity as a result (2, 149, 209). A comparative, phase III study conducted in 1992 reported that the combinatory therapy of Streptozocin + Doxorubicin provided more favorable results than Streptozocin + 5-FU in patients with advanced PNENs (210). However, the results described in this study have not been confirmed in any subsequent study (163, 209). Kouvaraki and colleagues retrospectively studied 84 PNEC patients treated with Streptozocin, 5-FU and Doxorubicin. Response rate was 39% with 2-year PFS and OS of 41% and 74%, respectively (211). Dacarbazine, a second alkylating agent, serves as a less toxic alternative. A phase II study tested Dacarbazine as a monotherapy in 50 PNEN patients and reported an ORR of 34% and median OS of 19.3 months (212). When combined with 5-FU, the overall response rate and PFS in advanced NENs were 38.2% and 13.9 months, respectively (213). A third alkylating agent that is primarily used as monotherapy for treatment of glioblastoma and melanoma is Temozolomide (163, 209). When combined with other compounds including Capecitabine (214), Bevacizumab (215), Bevacizumab and Octreotide (216), Thalidomide (217) and Everolimus (200) it shows significant activity in advanced PNENs (149, 209). A 2011 retrospective study reported that the combination treatment Capecitabine + Temozolomide (CAPTEM) in 30 chemonaive NEC patients resulted in an ORR of 70% with a PFS of 18 months (214). In 2018, a prospective, randomized phase II trial investigated Temozolomide therapy versus the CAPTEM combination therapy in PNEN patients. PFS was significantly better in the latter group (14.4 vs. 22.7 months) (218). However, a more recent retrospective analysis showed that CAPTEM was not able to improve PFS. Consequently, it was suggested by the authors that CAPTEM might be more useful for tumor shrinkage rather than improving PFS (219).
In poorly-differentiated G3 NECs, platinum agent regimens are often used in patients with adequate performance status. The first-line chemotherapy for NEC patients encompasses Cisplatin or Carboplatin combined with Etoposide or Irinotecan, based on the reported results (52, 220-223). Moertel and colleagues investigated the effect of Cisplatin + Etoposide in 45 patients with metastatic NENs, of which 27 were well-differentiated. The ORR was 67% in the 18 poorly-differentiated NECs with complete response (CR) in 17% of the patients, while unfortunately, only 2 patients (17%) of the well-differentiated NEN patients showed a response. Moreover, they reported a median survival of 19 months which was significantly longer than those described in literature (6-7 months). However, toxicity was a major issue (220). These results were confirmed by Mitry and colleagues in 1999 (221). Lower toxicity levels were observed when patients were treated with Carboplatin, but efficacy was similar to that of Cisplatin, rendering Carboplatin a valuable alternative (222, 224). Moreover, Oxaliplatin-based therapy appeared to have a greater activity in advanced PNETs (207).
The role of second-line chemotherapy for NEC patients is currently unknown, but many combinatory options have been examined (223, 225). Capecitabine + Oxaliplatin (CAPOX) and 5-FU + Oxaliplatin (FOLFOX) have been evaluated in two retrospective trials in well-differentiated NENs. ORRs of 26% and 30% were reported, respectively (226, 227). In addition, FOLFIRI and FOLFIRINOX (5-FU-based chemotherapies) have recently proven some effect in NEC patients progressing on platinum-based regimens (225, 228).
Radiotherapy
PEPTIDE RECEPTOR RADIONUCLIDE THERAPY (PRRT)
Peptide receptor radionuclide therapy (PRRT) is a therapy whereby a radiolabeled SSA (117Lutetium or 90Yttrium) is used to treat SSTR-positive, locally advanced and/or metastatic GEP-NENs, including PNENs. Adverse effects include nausea, renal toxicity, transient bone marrow suppression and seldom myelodysplastic syndrome or acute myeloid leukemia in 1-2% of patients (7, 149, 229, 230).
In a study of 504 patients, treatment with the analog 177Lu-DOTATATE showed activity in GEP-NENS (230). Looking specifically at the PNEN subgroup there was a 6% CR and a 36% PR in NF-PNENs and no CR and 47% PRs in functioning PNENs (230). Striking improvements in QOL of responders was also noted (231). A more recent study of 68 patients with PNENs treated with PRRT showed PRs in 41 patients (60.3%), minor responses in 8 (11.8%), SD in 9 (13.2%) and PD in 10 (14.7%) (232). The authors concluded that the outstanding response rates and survival outcomes suggest that PRRT is highly effective in advanced G1/2 PNENs when compared to other treatment modalities. Independent predictors of survival were the tumor proliferation index, the patient’s performance status, tumor burden and baseline plasma NSE level. PRRT also provided improvements in PFS compared to Octreotide in midgut NENs (232). The NETTER-1 phase III trial confirmed the efficacy in PRRT in midgut NENs in 2017 (229, 233, 234). Therefore, the FDA approved use of 177Lu-DOTATATE based on the results obtained in the NETTER-1 trial in midgut NETs (229, 234). Thus, the number of centers where this treatment is available is expected to increase in the US, although it has been used in Europe since 1996. Joint society practice guidelines have been developed (235). There are a number of ongoing international clinical trials listed on Clinical Trials.gov. Third party payer reimbursement is an ongoing issue which hopefully will be resolved.
For PNENs, the effects of PRRT have only been investigated in several single-arm prospective and retrospective trials (229). These, however, identified several signals in favor of PRRT use in PNENs as disease control rates and PFS varied between 84-85% and 30-34 months, respectively (232, 236). Additionally, a meta-analysis compared the efficiency of PRRT to Everolimus in GEP-NENs that were not eligible to surgical resection. An ORR of 47% was reported in the PRRT treated subgroup versus only 12% of the Everolimus treated patients. Moreover, disease control rates (81% vs. 73%) as well as PFS (25.7 vs. 14.7 months) were also superior in the PRRT treated subgroup (237). A recent retrospective study evaluated the association of upfront PRRT vs. upfront chemotherapy or targeted therapy with PFS in enteropancreatic NET patients who progressed under SSA treatment. Patients with a Ki-67 value of ≤ 10% who received upfront PRRT, were reported to have a statically and clinically meaningful prolonged PFS (238). Based on these findings, it seems important to better define the role of PRRT in the treatment of PNENs within the future.
Liver-Directed Therapy
As mentioned earlier, the liver constitutes the most common site for distant metastases (52, 172-174). Since surgical resection and RFA are only feasible in a minority of patients, there are multiple liver-directed therapies available to treat the remaining patients. These methods include transarterial chemoembolization (TACE), transarterial embolization (TAE), or radioembolization, which will be discussed below. Given the lack of randomized data, it is difficult to determine with certainty which method is preferred. Moreover, NANETS guidelines recommend to consider systemic therapy rather than liver-directed therapy when >50-75% hepatic tumor burden is present (239).
A study of chemoembolization combined with SSA treatment resulted in a relief of symptoms in 78% of the patients. Monitoring serum pancreastatin levels predicted a response to this therapy in which radiographic improvement or stability were seen in 45% of patients (77). In NEN patients that underwent TACE, plasma levels of pancreastatin above 5000 pg/ml pre-treatment were associated with increased peri-procedure mortality (240).
Radioembolization (also known as selective intrahepatic radiotherapy or SIRT) involves embolization of 90Yttrium embedded either in a resin microsphere (Sir-Sphere) or a glass microsphere (TheraSphere). Acute toxicities associated with 90Yttrium microsphere embolization appear to be lower than other embolization techniques, primarily due to the fact that the procedure does not induce ischemic hepatitis. Thus, the procedure can be performed on an outpatient basis. A rare, but potentially serious complication is radiation enteritis, which can occur if particles are accidentally infused into arteries supplying the gastrointestinal tract. Chronic radiation hepatitis is another potential toxicity. Response rates associated with radioembolization in metastatic NEN patients have been encouraging. In one retrospective multi-center study of 148 patients treated with Sir-Spheres, the objective radiographic response rate was 63% with a median survival of 70 months, with no radiation-induced liver failure (241). Another study of 42 patients treated with either Sir-Spheres or TheraSpheres reported a response rate of 51%. However, only 29 of the 42 enrolled patients were evaluable (242). Grozinsky-Glasberg and colleagues examined 57 patients which underwent either TACE, TAE or SIRT. They reported symptomatic control and a stabilization of tumor growth in 95% of the patients. Noteworthy, they observed improvements regardless of the extent of the liver metastasis (243).
Novel Targets for the Treatment of (P)NENs
While there has been a quantum leap in the ability to treat NENs successfully we have a long way to go to cure the disease. Fortunately, research into improved and novel therapeutic strategies is ongoing. So far, the results of immunotherapy as monotherapy in PNET patients remain disappointing. Examples include the inhibition of the programmed death-ligand 1 (PDL-1) and cytotoxic T-lymphocyte antigen-4 (CTLA4), by treating patients with Pembrolizumab to enhance the immune response towards tumor cells (186, 244, 245). The KEYNOTE-028 phase I study treated PDL-1 positive PNEN patients with Pembrolizumab and reported an ORR of 6.3%, but no CRs occurred. Responses were better in metastatic carcinoids (ORR: 12%) (246). These findings were confirmed by the KEYNOTE-158 phase II study, in which the ORR was 3.7% with 3 PRs in PNEN and 1 in rectal NEN patients (247). Currently, several other clinical trials that are investigating the antitumor effect of immune checkpoint inhibitors include NCT02939651 for Pembrolizumab and NCT02955069 for other PDL-1 receptor antibodies (247). Moreover, there is also much speculation that PRRT cytotoxic drugs induce genotoxicity, hence increase the neoantigen load and thereby could potentially enhance the efficacy of immunotherapy (149, 244, 245, 248). Bevacizumab, a monoclonal antibody against the VEGF, showed no real benefit in PFS in a phase III trial in which Bevacizumab + Octreotide was compared to Interferon + Octreotide (249). The BETTER phase II trial, on the other hand, demonstrated that Bevacizumab + 5-FU/Streptozocin in patients with metastatic, well-differentiated PNENs could reach a PFS of 23.7 months and they reported an OS at 24 months of 88% (250). The SANET-ep (251) and SANET-p (252) phase III studies examined the efficacy and safety of Surufatinib in extrapancreatic NENs and PNENs, respectively. Surufatinib, a small-molecule inhibitor that targets VEGFRs as well as the fibroblast growth factor (FGF) receptor 1 and macrophage colony-stimulating factor 1 (CSF1) receptor, effectively prolonged PFS in both studies and was therefore suggested a potential treatment option in both patient populations (251, 252).
QUALITY OF LIFE IN PATIENTS WITH PNENS
The measurement of health-related quality of life (HRQOL) has become essential for evaluating the impact of the disease process and the treatment on patient’s symptoms, social, emotional, physiological, and physical functioning. The European Organization for Research and Treatment of Cancer (EORTC) developed the QLQ-C30 tool for oncology patients (253) and the QLQ-GINET21 tool was specifically developed for a spectrum of NEN patients (28% PNENs) (254). The Norfolk QOLNET was specifically developed for midgut NETs (carcinoids) and may provide some additional advantages for that specific group of patients (231).
The most commonly used QOL tool in GEP-NENs (including PNENs) is the EORTC QLQ-C30 (255). SSAs and Sunitinib have shown to improve HRQOL in diverse groups of GEP-NEN patients (255). In the CLARINET study, QLC-C30 data were mapped to EQ-5D utilities and not surprisingly worse utility values were noted with PD compared to SD. Of note, tumor location (midgut vs. pancreas) did not affect utility (256). PNEN patients treated with everolimus showed stable HRQOL scores as opposed to worse scores in non-PNEN patients (Pavel). PRRT treatment of PNEN patients resulted in significantly improved global health status, social functioning and mitigation of physical complaints (257). Thus, data are emerging on HRQOL in PNEN patients. However, most studies are too heterogenous in terms of patient populations and treatment interventions to draw firm conclusions (258). Moving forward, it will be important for HRQOL to be measured as a key component of clinical trials.
EXPERT COMMENTARY
After many years of frustration, our knowledge regarding the biology, pathophysiology and genetics of (P)NENs increased. This has led to marked improvements in (functional) imaging, with the development of 68Gallium-labeled SSAs, as well as targeted treatments such as the tyrosine-kinase inhibitor Sunitinib, and the mTOR inhibitor Everolimus. In addition, PRRT seems to be expanding its role in treatment from midgut to PNENs. In the future, both imaging and treatment options will continue to evolve as more specific imaging agents and therapeutic targets are being developed and evaluated in numerous studies. The relatively uncommon nature of PNENs has made designing and completing randomized studies of adequate power challenging for a single institution. Therefore, we encourage the recent trend of multi-institutional, multinational studies in more homogeneous patient populations. We also strongly agree with the recommendation of NANETS, ENETS and other groups that all of these patients should be entered into clinical trials whenever feasible. Determining study availability and patient eligibility has been greatly facilitated by Clinical trials.gov as well as institutional and organizational websites. Enrolling more patients in clinical trials by overcoming barriers to participation will be required to move patient care forward.
Unfortunately, to date, the optimal treatment(s) and treatment sequences have yet to be defined. The lack of treatment standardization, the plethora of treatments that most patients receive, and different treatment sequences make it extremely difficult to assess the effectiveness of a particular treatment relative to others. Moreover, head-to-head comparisons are lacking as well. Available consensus guidelines establish broad principles, but are generally not helpful in managing a specific patient. Management has become even more complex given the multiplicity of effective treatments for advanced disease, none of which has convincingly been shown to be superior to the other. Hence, an experienced multidisciplinary team is essential to guide management of these patients. Given relative parity of effectiveness, decisions regarding choice of treatment need to be based on multiple considerations, including patient’s overall health, disease burden, symptomatology, rate of progression, treatment toxicity, effect on QOL, and cost. These considerations will usually lead to one treatment being favored over another.
Because of the relatively indolent nature of many (or most) NENs, long-term follow-up to assess differences in treatment outcomes, is required. However, biomarkers that can predict response to a particular therapy are currently not available. We expect that in the future the so-called tumor circulome, especially ctDNA, could play an important role in this as recent studies revealed its potential to diagnose, prognosticate and monitor disease progression.
5. Year View
Knowledge of the biology and genetics will continue to accumulate, which could potentially lead to further refinements in classification, staging, and personalized treatment. Genetic profiling will become clinically useful as data will accumulate on treatment effectiveness in patient subgroups leading to more tailored therapies. Moreover, biomarkers will be developed that better predict response to a particular therapy. Results of the ongoing clinical trials on newer SSAs and targeted agents will add to the number of available treatments. The role of PRRT in the treatment of PNENs will be better defined. There will be increased knowledge as to optimal treatment sequences. Designing randomized clinical trials of adequate power will remain a challenge for many reasons including the scarcity and indolent growth of these tumors. Consensus guidelines will evolve, but patient management will continue to require an experienced multidisciplinary team.
REFERENCES
- 1.
- Clift AK, Kidd M, Bodei L, Toumpanakis C, Baum RP, Oberg K, et al. Neuroendocrine Neoplasms of the Small Bowel and Pancreas. Neuroendocrinology. 2020;110(6):444–76. [PMC free article: PMC9175236] [PubMed: 31557758]
- 2.
- Metz DC, Jensen RT. Gastrointestinal neuroendocrine tumors: pancreatic endocrine tumors. Gastroenterology. 2008;135(5):1469–92. [PMC free article: PMC2612755] [PubMed: 18703061]
- 3.
- Perri G, Prakash LR, Katz MHG. Pancreatic neuroendocrine tumors. Current opinion in gastroenterology. 2019;35(5):468–77. [PubMed: 31306159]
- 4.
- Hong X, Qiao S, Li F, Wang W, Jiang R, Wu H, et al. Whole-genome sequencing reveals distinct genetic bases for insulinomas and non-functional pancreatic neuroendocrine tumours: leading to a new classification system. Gut. 2020;69(5):877–87. [PMC free article: PMC7229893] [PubMed: 31462556]
- 5.
- Dasari A, Shen C, Halperin D, Zhao B, Zhou S, Xu Y, et al. Trends in the Incidence, Prevalence, and Survival Outcomes in Patients With Neuroendocrine Tumors in the United States. JAMA oncology. 2017;3(10):1335–42. [PMC free article: PMC5824320] [PubMed: 28448665]
- 6.
- Daskalakis K. Functioning and nonfunctioning pNENs. Current Opinion in Endocrine and Metabolic Research. 2021;18:284–90.
- 7.
- Ma ZY, Gong YF, Zhuang HK, Zhou ZX, Huang SZ, Zou YP, et al. Pancreatic neuroendocrine tumors: A review of serum biomarkers, staging, and management. World journal of gastroenterology. 2020;26(19):2305–22. [PMC free article: PMC7243647] [PubMed: 32476795]
- 8.
- Cives M, Strosberg JR. Gastroenteropancreatic Neuroendocrine Tumors. CA: a cancer journal for clinicians. 2018;68(6):471–87. [PubMed: 30295930]
- 9.
- Falconi M, Eriksson B, Kaltsas G, Bartsch DK, Capdevila J, Caplin M, et al. ENETS Consensus Guidelines Update for the Management of Patients with Functional Pancreatic Neuroendocrine Tumors and Non-Functional Pancreatic Neuroendocrine Tumors. Neuroendocrinology. 2016;103(2):153–71. [PMC free article: PMC4849884] [PubMed: 26742109]
- 10.
- Boons G, Vandamme T, Peeters M, Van Camp G, Op de Beeck K. Clinical applications of (epi)genetics in gastroenteropancreatic neuroendocrine neoplasms: Moving towards liquid biopsies. Reviews in endocrine & metabolic disorders. 2019;20(3):333–51. [PubMed: 31368038]
- 11.
- Liakakos T, Roukos DH. Everolimus and sunitinib: from mouse models to treatment of pancreatic neuroendocrine tumors. Future oncology (London, England). 2011;7(9):1025–9. [PubMed: 21919689]
- 12.
- Vinik AI. Advances in diagnosis and treatment of pancreatic neuroendocrine tumors. Endocrine practice : official journal of the American College of Endocrinology and the American Association of Clinical Endocrinologists. 2014;20(11):1222-30. [PubMed: 25297671]
- 13.
- Verbeke CS. Endocrine tumours of the pancreas. Histopathology. 2010;56(6):669–82. [PubMed: 20546332]
- 14.
- Plöckinger U, Wiedenmann B. Diagnosis of non-functioning neuro-endocrine gastro-enteropancreatic tumours. Neuroendocrinology. 2004;80 Suppl 1:35–8. [PubMed: 15477715]
- 15.
- Inzani F, Petrone G, Rindi G. The New World Health Organization Classification for Pancreatic Neuroendocrine Neoplasia. Endocrinology and metabolism clinics of North America. 2018;47(3):463–70. [PubMed: 30098710]
- 16.
- Nagtegaal ID, Odze RD, Klimstra D, Paradis V, Rugge M, Schirmacher P, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76(2):182–8. [PMC free article: PMC7003895] [PubMed: 31433515]
- 17.
- Ciobanu OA, Martin S, Fica S. Perspectives on the diagnostic, predictive and prognostic markers of neuroendocrine neoplasms. Experimental and therapeutic medicine. 2021;22(6):1479. (Review) [PMC free article: PMC8576627] [PubMed: 34765020]
- 18.
- Jensen RT, Berna MJ, Bingham DB, Norton JA. Inherited pancreatic endocrine tumor syndromes: advances in molecular pathogenesis, diagnosis, management, and controversies. Cancer. 2008;113(7) Suppl:1807–43. [PMC free article: PMC2574000] [PubMed: 18798544]
- 19.
- Li X, Gou S, Liu Z, Ye Z, Wang C. Assessment of the American Joint Commission on Cancer 8th Edition Staging System for Patients with Pancreatic Neuroendocrine Tumors: A Surveillance, Epidemiology, and End Results analysis. Cancer medicine. 2018;7(3):626-34. [PMC free article: PMC5852336] [PubMed: 29380547]
- 20.
- You Y, Jang JY, Kim SC, Yoon YS, Park JS, Cho CK, et al. Validation of the 8th AJCC Cancer Staging System for Pancreas Neuroendocrine Tumors Using Korean Nationwide Surgery Database. Cancer research and treatment. 2019;51(4):1639–52. [PMC free article: PMC6790839] [PubMed: 30999719]
- 21.
- Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, et al. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more "personalized" approach to cancer staging. CA: a cancer journal for clinicians. 2017;67(2):93-9. [PubMed: 28094848]
- 22.
- Yang M, Zeng L, Yao WQ, Ke NW, Tan CL, Tian BL, et al. A comprehensive validation of the novel 8th edition of American Joint Committee on Cancer staging manual for the long-term survivals of patients with non-functional pancreatic neuroendocrine neoplasms. Medicine. 2020;99(46):e22291. [PMC free article: PMC7668515] [PubMed: 33181635]
- 23.
- Reid MD, Balci S, Saka B, Adsay NV. Neuroendocrine tumors of the pancreas: current concepts and controversies. Endocrine pathology. 2014;25(1):65–79. [PubMed: 24430597]
- 24.
- Di Domenico A, Pipinikas CP, Maire RS, Bräutigam K, Simillion C, Dettmer MS, et al. Epigenetic landscape of pancreatic neuroendocrine tumours reveals distinct cells of origin and means of tumour progression. Communications biology. 2020;3(1):740. [PMC free article: PMC7721725] [PubMed: 33288854]
- 25.
- Noone TC, Hosey J, Firat Z, Semelka RC. Imaging and localization of islet-cell tumours of the pancreas on CT and MRI. Best practice & research Clinical endocrinology & metabolism. 2005;19(2):195–211. [PubMed: 15763695]
- 26.
- Taskin OC, Clarke CN, Erkan M, Tsai S, Evans DB, Adsay V. Pancreatic neuroendocrine neoplasms: current state and ongoing controversies on terminology, classification and prognostication. Journal of gastrointestinal oncology. 2020;11(3):548–58. [PMC free article: PMC7340800] [PubMed: 32655934]
- 27.
- Gao HL, Wang WQ, Yu XJ, Liu L. Molecular drivers and cells of origin in pancreatic ductal adenocarcinoma and pancreatic neuroendocrine carcinoma. Experimental hematology & oncology. 2020;9:28. [PMC free article: PMC7579802] [PubMed: 33101770]
- 28.
- Chan CS, Laddha SV, Lewis PW, Koletsky MS, Robzyk K, Da Silva E, et al. ATRX, DAXX or MEN1 mutant pancreatic neuroendocrine tumors are a distinct alpha-cell signature subgroup. Nature communications. 2018;9(1):4158. [PMC free article: PMC6185985] [PubMed: 30315258]
- 29.
- Cejas P, Drier Y, Dreijerink KMA, Brosens LAA, Deshpande V, Epstein CB, et al. Enhancer signatures stratify and predict outcomes of non-functional pancreatic neuroendocrine tumors. Nature medicine. 2019;25(8):1260–5. [PMC free article: PMC6919319] [PubMed: 31263286]
- 30.
- Strobel O, Rosow DE, Rakhlin EY, Lauwers GY, Trainor AG, Alsina J, et al. Pancreatic duct glands are distinct ductal compartments that react to chronic injury and mediate Shh-induced metaplasia. Gastroenterology. 2010;138(3):1166–77. [PMC free article: PMC3806111] [PubMed: 20026066]
- 31.
- Anderson CW, Bennett JJ. Clinical Presentation and Diagnosis of Pancreatic Neuroendocrine Tumors. Surgical oncology clinics of North America. 2016;25(2):363–74. [PubMed: 27013370]
- 32.
- Mimeault M, Batra SK. Recent progress on normal and malignant pancreatic stem/progenitor cell research: therapeutic implications for the treatment of type 1 or 2 diabetes mellitus and aggressive pancreatic cancer. Gut. 2008;57(10):1456–68. [PMC free article: PMC2836486] [PubMed: 18791122]
- 33.
- Jiang FX, Morahan G. Multipotent pancreas progenitors: Inconclusive but pivotal topic. World journal of stem cells. 2015;7(11):1251–61. [PMC free article: PMC4691693] [PubMed: 26730269]
- 34.
- Zhong F, Jiang Y. Endogenous Pancreatic β Cell Regeneration: A Potential Strategy for the Recovery of β Cell Deficiency in Diabetes. Frontiers in endocrinology. 2019;10:101. [PMC free article: PMC6391341] [PubMed: 30842756]
- 35.
- Yamaguchi J, Liss AS, Sontheimer A, Mino-Kenudson M, Castillo CF, Warshaw AL, et al. Pancreatic duct glands (PDGs) are a progenitor compartment responsible for pancreatic ductal epithelial repair. Stem cell research. 2015;15(1):190–202. [PMC free article: PMC5396061] [PubMed: 26100232]
- 36.
- Tirosh A, Kebebew E. Genetic and epigenetic alterations in pancreatic neuroendocrine tumors. Journal of gastrointestinal oncology. 2020;11(3):567–77. [PMC free article: PMC7340809] [PubMed: 32655936]
- 37.
- Görtz B, Roth J, Krähenmann A, de Krijger RR, Muletta-Feurer S, Rütimann K, et al. Mutations and allelic deletions of the MEN1 gene are associated with a subset of sporadic endocrine pancreatic and neuroendocrine tumors and not restricted to foregut neoplasms. The American journal of pathology. 1999;154(2):429–36. [PMC free article: PMC1849990] [PubMed: 10027401]
- 38.
- Mafficini A, Scarpa A. Genetics and Epigenetics of Gastroenteropancreatic Neuroendocrine Neoplasms. Endocrine reviews. 2019;40(2):506–36. [PMC free article: PMC6534496] [PubMed: 30657883]
- 39.
- Scarpa A, Chang DK, Nones K, Corbo V, Patch AM, Bailey P, et al. Whole-genome landscape of pancreatic neuroendocrine tumours. Nature. 2017;543(7643):65–71. [PubMed: 28199314]
- 40.
- Crona J, Skogseid B. GEP- NETS UPDATE: Genetics of neuroendocrine tumors. European journal of endocrinology. 2016;174(6):R275–90. [PubMed: 27165966]
- 41.
- Jiao Y, Shi C, Edil BH, de Wilde RF, Klimstra DS, Maitra A, et al. DAXX/ATRX, MEN1, and mTOR pathway genes are frequently altered in pancreatic neuroendocrine tumors. Science (New York, NY). 2011;331(6021):1199–203. [PMC free article: PMC3144496] [PubMed: 21252315]
- 42.
- Gläsker S, Vergauwen E, Koch CA, Kutikov A, Vortmeyer AO. Von Hippel-Lindau Disease: Current Challenges and Future Prospects. OncoTargets and therapy. 2020;13:5669–90. [PMC free article: PMC7305855] [PubMed: 32606780]
- 43.
- Zwolak A, Świrska J, Tywanek E, Dudzińska M, Tarach JS, Matyjaszek-Matuszek B. Pancreatic neuroendocrine tumours in patients with von Hippel-Lindau disease. Endokrynologia Polska. 2020;71(3):256–9. [PubMed: 32797472]
- 44.
- Parekh VI, Modali SD, Welch J, Simonds WF, Weinstein LS, Kebebew E, et al. Frequency and consequence of the recurrent YY1 p.T372R mutation in sporadic insulinomas. Endocrine-related cancer. 2018;25(5):L31–l5. [PMC free article: PMC5862779] [PubMed: 29467239]
- 45.
- Pipinikas CP, Berner AM, Sposito T, Thirlwell C. The evolving (epi)genetic landscape of pancreatic neuroendocrine tumours. Endocrine-related cancer. 2019;26(9):R519–r44. [PubMed: 31252410]
- 46.
- Singhi AD, Klimstra DS. Well-differentiated pancreatic neuroendocrine tumours (PanNETs) and poorly differentiated pancreatic neuroendocrine carcinomas (PanNECs): concepts, issues and a practical diagnostic approach to high-grade (G3) cases. Histopathology. 2018;72(1):168–77. [PubMed: 29239037]
- 47.
- Boons G, Vandamme T, Mariën L, Lybaert W, Roeyen G, Rondou T, et al. Longitudinal Copy-Number Alteration Analysis in Plasma Cell-Free DNA of Neuroendocrine Neoplasms is a Novel Specific Biomarker for Diagnosis, Prognosis, and Follow-up. Clinical cancer research : an official journal of the American Association for Cancer Research. 2021 [PMC free article: PMC9401546] [PubMed: 34759042]
- 48.
- House MG, Herman JG, Guo MZ, Hooker CM, Schulick RD, Lillemoe KD, et al. Aberrant hypermethylation of tumor suppressor genes in pancreatic endocrine neoplasms. Annals of surgery. 2003;238(3):423–31. [PMC free article: PMC1422710] [PubMed: 14501508]
- 49.
- Neiman D, Moss J, Hecht M, Magenheim J, Piyanzin S, Shapiro AMJ, et al. Islet cells share promoter hypomethylation independently of expression, but exhibit cell-type-specific methylation in enhancers. Proceedings of the National Academy of Sciences of the United States of America. 2017;114(51):13525–30. [PMC free article: PMC5754795] [PubMed: 29203669]
- 50.
- Boons G, Vandamme T, Ibrahim J, Roeyen G, Driessen A, Peeters D, et al. PDX1 DNA Methylation Distinguishes Two Subtypes of Pancreatic Neuroendocrine Neoplasms with a Different Prognosis. Cancers. 2020;12(6) [PMC free article: PMC7352978] [PubMed: 32512761]
- 51.
- Lakis V, Lawlor RT, Newell F, Patch AM, Mafficini A, Sadanandam A, et al. DNA methylation patterns identify subgroups of pancreatic neuroendocrine tumors with clinical association. Communications biology. 2021;4(1):155. [PMC free article: PMC7859232] [PubMed: 33536587]
- 52.
- Scott AT, Howe JR. Evaluation and Management of Neuroendocrine Tumors of the Pancreas. The Surgical clinics of North America. 2019;99(4):793–814. [PMC free article: PMC6601637] [PubMed: 31255207]
- 53.
- Lee DW, Kim MK, Kim HG. Diagnosis of Pancreatic Neuroendocrine Tumors. Clinical endoscopy. 2017;50(6):537–45. [PMC free article: PMC5719919] [PubMed: 29207856]
- 54.
- Rebours V, Cordova J, Couvelard A, Fabre M, Palazzo L, Vullierme MP, et al. Can pancreatic neuroendocrine tumour biopsy accurately determine pathological characteristics? Digestive and liver disease : official journal of the Italian Society of Gastroenterology and the Italian Association for the Study of the Liver. 2015;47(11):973-7. [PubMed: 26169284]
- 55.
- Klöppel G, Couvelard A, Perren A, Komminoth P, McNicol AM, Nilsson O, et al. ENETS Consensus Guidelines for the Standards of Care in Neuroendocrine Tumors: towards a standardized approach to the diagnosis of gastroenteropancreatic neuroendocrine tumors and their prognostic stratification. Neuroendocrinology. 2009;90(2):162–6. [PubMed: 19060454]
- 56.
- Eusebi LH, Thorburn D, Toumpanakis C, Frazzoni L, Johnson G, Vessal S, et al. Endoscopic ultrasound-guided fine-needle aspiration vs fine-needle biopsy for the diagnosis of pancreatic neuroendocrine tumors. Endoscopy international open. 2019;7(11):E1393–e9. [PMC free article: PMC6805236] [PubMed: 31673610]
- 57.
- Crinò SF, Ammendola S, Meneghetti A, Bernardoni L, Conti Bellocchi MC, Gabbrielli A, et al. Comparison between EUS-guided fine-needle aspiration cytology and EUS-guided fine-needle biopsy histology for the evaluation of pancreatic neuroendocrine tumors. Pancreatology : official journal of the International Association of Pancreatology (IAP) [et al]. 2021;21(2):443-50. [PubMed: 33390343]
- 58.
- Garcia-Carbonero R, Sorbye H, Baudin E, Raymond E, Wiedenmann B, Niederle B, et al. ENETS Consensus Guidelines for High-Grade Gastroenteropancreatic Neuroendocrine Tumors and Neuroendocrine Carcinomas. Neuroendocrinology. 2016;103(2):186–94. [PubMed: 26731334]
- 59.
- Fang JM, Shi J. A Clinicopathologic and Molecular Update of Pancreatic Neuroendocrine Neoplasms With a Focus on the New World Health Organization Classification. Archives of pathology & laboratory medicine. 2019;143(11):1317–26. [PMC free article: PMC7141760] [PubMed: 31509453]
- 60.
- Perren A, Couvelard A, Scoazec JY, Costa F, Borbath I, Delle Fave G, et al. ENETS Consensus Guidelines for the Standards of Care in Neuroendocrine Tumors: Pathology: Diagnosis and Prognostic Stratification. Neuroendocrinology. 2017;105(3):196–200. [PubMed: 28190015]
- 61.
- Rockall AG, Reznek RH. Imaging of neuroendocrine tumours (CT/MR/US). Best practice & research Clinical endocrinology & metabolism. 2007;21(1):43–68. [PubMed: 17382265]
- 62.
- Virgolini I, Traub-Weidinger T, Decristoforo C. Nuclear medicine in the detection and management of pancreatic islet-cell tumours. Best practice & research Clinical endocrinology & metabolism. 2005;19(2):213–27. [PubMed: 15763696]
- 63.
- Gibril F, Jensen RT. Diagnostic uses of radiolabelled somatostatin receptor analogues in gastroenteropancreatic endocrine tumours. Digestive and liver disease : official journal of the Italian Society of Gastroenterology and the Italian Association for the Study of the Liver. 2004;36 Suppl 1:S106-20. [PubMed: 15077919]
- 64.
- Sundin A, Garske U, Orlefors H. Nuclear imaging of neuroendocrine tumours. Best practice & research Clinical endocrinology & metabolism. 2007;21(1):69–85. [PubMed: 17382266]
- 65.
- Singh A, Hines JJ, Friedman B. Multimodality Imaging of the Pancreatic Neuroendocrine Tumors. Seminars in Ultrasound. CT and MRI. 2019;40(6):469–82. [PubMed: 31806146]
- 66.
- Chiti G, Grazzini G, Cozzi D, Danti G, Matteuzzi B, Granata V, et al. Imaging of Pancreatic Neuroendocrine Neoplasms. International journal of environmental research and public health. 2021;18(17) [PMC free article: PMC8430610] [PubMed: 34501485]
- 67.
- Morse B, Al-Toubah T, Montilla-Soler J. Anatomic and Functional Imaging of Neuroendocrine Tumors. Current treatment options in oncology. 2020;21(9):75. [PubMed: 32728967]
- 68.
- Khanna L, Prasad SR, Sunnapwar A, Kondapaneni S, Dasyam A, Tammisetti VS, et al. Pancreatic Neuroendocrine Neoplasms: 2020 Update on Pathologic and Imaging Findings and Classification. Radiographics : a review publication of the Radiological Society of North America, Inc. 2020;40(5):1240–62. [PubMed: 32795239]
- 69.
- Rozenblum L, Mokrane FZ, Yeh R, Sinigaglia M, Besson FL, Seban RD, et al. Imaging-guided precision medicine in non-resectable gastro-entero-pancreatic neuroendocrine tumors: A step-by-step approach. European journal of radiology. 2020;122:108743. [PubMed: 31783345]
- 70.
- Galgano SJ, Sharbidre K, Morgan DE. Multimodality Imaging of Neuroendocrine Tumors. Radiologic clinics of North America. 2020;58(6):1147–59. [PubMed: 33040854]
- 71.
- Tamm EP, Bhosale P, Lee JH, Rohren EM. State-of-the-art Imaging of Pancreatic Neuroendocrine Tumors. Surgical oncology clinics of North America. 2016;25(2):375–400. [PMC free article: PMC4808582] [PubMed: 27013371]
- 72.
- Sundin A, Vullierme MP, Kaltsas G, Plöckinger U. ENETS Consensus Guidelines for the Standards of Care in Neuroendocrine Tumors: radiological examinations. Neuroendocrinology. 2009;90(2):167–83. [PubMed: 19077417]
- 73.
- Jackson JE. Angiography and arterial stimulation venous sampling in the localization of pancreatic neuroendocrine tumours. Best practice & research Clinical endocrinology & metabolism. 2005;19(2):229–39. [PubMed: 15763697]
- 74.
- McLean AM, Fairclough PD. Endoscopic ultrasound in the localisation of pancreatic islet cell tumours. Best practice & research Clinical endocrinology & metabolism. 2005;19(2):177–93. [PubMed: 15763694]
- 75.
- Norton JA, Jensen RT. Resolved and unresolved controversies in the surgical management of patients with Zollinger-Ellison syndrome. Annals of surgery. 2004;240(5):757–73. [PMC free article: PMC1356480] [PubMed: 15492556]
- 76.
- Krenning EP, Kwekkeboom DJ, Oei HY, de Jong RJ, Dop FJ, Reubi JC, et al. Somatostatin-receptor scintigraphy in gastroenteropancreatic tumors. An overview of European results. Annals of the New York Academy of Sciences. 1994;733:416–24. [PubMed: 7978890]
- 77.
- Desai H, Borges-Neto S, Wong TZ. Molecular Imaging and Therapy for Neuroendocrine Tumors. Current treatment options in oncology. 2019;20(10):78. [PubMed: 31468190]
- 78.
- Termanini B, Gibril F, Reynolds JC, Doppman JL, Chen CC, Stewart CA, et al. Value of somatostatin receptor scintigraphy: a prospective study in gastrinoma of its effect on clinical management. Gastroenterology. 1997;112(2):335–47. [PubMed: 9024287]
- 79.
- Gibril F, Reynolds JC, Chen CC, Yu F, Goebel SU, Serrano J, et al. Specificity of somatostatin receptor scintigraphy: a prospective study and effects of false-positive localizations on management in patients with gastrinomas. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 1999;40(4):539-53. [PubMed: 10210211]
- 80.
- Christ E, Wild D, Ederer S, Béhé M, Nicolas G, Caplin ME, et al. Glucagon-like peptide-1 receptor imaging for the localisation of insulinomas: a prospective multicentre imaging study. The lancet Diabetes & endocrinology. 2013;1(2):115–22. [PubMed: 24622317]
- 81.
- Panagiotidis E, Alshammari A, Michopoulou S, Skoura E, Naik K, Maragkoudakis E, et al. Comparison of the Impact of 68Ga-DOTATATE and 18F-FDG PET/CT on Clinical Management in Patients with Neuroendocrine Tumors. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2017;58(1):91-6. [PubMed: 27516446]
- 82.
- Maxwell JE, Howe JR. Imaging in neuroendocrine tumors: an update for the clinician. International journal of endocrine oncology. 2015;2(2):159–68. [PMC free article: PMC4526141] [PubMed: 26257863]
- 83.
- Johnbeck CB, Knigge U, Kjær A. PET tracers for somatostatin receptor imaging of neuroendocrine tumors: current status and review of the literature. Future oncology (London, England). 2014;10(14):2259–77. [PubMed: 25471038]
- 84.
- Kornaczewski Jackson ER, Pointon OP, Bohmer R, Burgess JR. Utility of FDG-PET Imaging for Risk Stratification of Pancreatic Neuroendocrine Tumors in MEN1. The Journal of clinical endocrinology and metabolism. 2017;102(6):1926–33. [PubMed: 28323985]
- 85.
- Orlefors H, Sundin A, Garske U, Juhlin C, Oberg K, Skogseid B, et al. Whole-body (11)C-5-hydroxytryptophan positron emission tomography as a universal imaging technique for neuroendocrine tumors: comparison with somatostatin receptor scintigraphy and computed tomography. The Journal of clinical endocrinology and metabolism. 2005;90(6):3392–400. [PubMed: 15755858]
- 86.
- Gabriel M, Decristoforo C, Kendler D, Dobrozemsky G, Heute D, Uprimny C, et al. 68Ga-DOTA-Tyr3-octreotide PET in neuroendocrine tumors: comparison with somatostatin receptor scintigraphy and CT. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2007;48(4):508-18. [PubMed: 17401086]
- 87.
- Eriksson B, Orlefors H, Oberg K, Sundin A, Bergström M, Långström B. Developments in PET for the detection of endocrine tumours. Best practice & research Clinical endocrinology & metabolism. 2005;19(2):311–24. [PubMed: 15763703]
- 88.
- Geijer H, Breimer LH. Somatostatin receptor PET/CT in neuroendocrine tumours: update on systematic review and meta-analysis. European journal of nuclear medicine and molecular imaging. 2013;40(11):1770–80. [PubMed: 23873003]
- 89.
- Deppen SA, Liu E, Blume JD, Clanton J, Shi C, Jones-Jackson LB, et al. Safety and Efficacy of 68Ga-DOTATATE PET/CT for Diagnosis, Staging, and Treatment Management of Neuroendocrine Tumors. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2016;57(5):708-14. [PMC free article: PMC5362940] [PubMed: 26769865]
- 90.
- Sadowski SM, Neychev V, Millo C, Shih J, Nilubol N, Herscovitch P, et al. Prospective Study of 68Ga-DOTATATE Positron Emission Tomography/Computed Tomography for Detecting Gastro-Entero-Pancreatic Neuroendocrine Tumors and Unknown Primary Sites. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2016;34(6):588–96. [PMC free article: PMC4872030] [PubMed: 26712231]
- 91.
- Etchebehere EC, de Oliveira Santos A, Gumz B, Vicente A, Hoff PG, Corradi G, et al. 68Ga-DOTATATE PET/CT, 99mTc-HYNIC-octreotide SPECT/CT, and whole-body MR imaging in detection of neuroendocrine tumors: a prospective trial. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2014;55(10):1598-604. [PubMed: 25168627]
- 92.
- Skoura E, Michopoulou S, Mohmaduvesh M, Panagiotidis E, Al Harbi M, Toumpanakis C, et al. The Impact of 68Ga-DOTATATE PET/CT Imaging on Management of Patients with Neuroendocrine Tumors: Experience from a National Referral Center in the United Kingdom. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2016;57(1):34-40. [PubMed: 26471695]
- 93.
- Crown A, Rocha FG, Raghu P, Lin B, Funk G, Alseidi A, et al. Impact of initial imaging with gallium-68 dotatate PET/CT on diagnosis and management of patients with neuroendocrine tumors. Journal of surgical oncology. 2020;121(3):480–5. [PubMed: 31853990]
- 94.
- Hofman MS, Lau WF, Hicks RJ. Somatostatin receptor imaging with 68Ga DOTATATE PET/CT: clinical utility, normal patterns, pearls, and pitfalls in interpretation. Radiographics : a review publication of the Radiological Society of North America, Inc. 2015;35(2):500–16. [PubMed: 25763733]
- 95.
- Lastoria S, Marciello F, Faggiano A, Aloj L, Caracò C, Aurilio M, et al. Role of (68)Ga-DOTATATE PET/CT in patients with multiple endocrine neoplasia type 1 (MEN1). Endocrine. 2016;52(3):488–94. [PubMed: 26242621]
- 96.
- Luo Y, Pan Q, Yao S, Yu M, Wu W, Xue H, et al. Glucagon-Like Peptide-1 Receptor PET/CT with 68Ga-NOTA-Exendin-4 for Detecting Localized Insulinoma: A Prospective Cohort Study. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2016;57(5):715-20. [PMC free article: PMC5227553] [PubMed: 26795291]
- 97.
- Elazezy M, Joosse SA. Techniques of using circulating tumor DNA as a liquid biopsy component in cancer management. Computational and structural biotechnology journal. 2018;16:370–8. [PMC free article: PMC6197739] [PubMed: 30364656]
- 98.
- Siravegna G, Marsoni S, Siena S, Bardelli A. Integrating liquid biopsies into the management of cancer. Nature reviews Clinical oncology. 2017;14(9):531–48. [PubMed: 28252003]
- 99.
- De Rubis G, Rajeev Krishnan S, Bebawy M. Liquid Biopsies in Cancer Diagnosis, Monitoring, and Prognosis. Trends in pharmacological sciences. 2019;40(3):172–86. [PubMed: 30736982]
- 100.
- Heitzer E, Haque IS, Roberts CES, Speicher MR. Current and future perspectives of liquid biopsies in genomics-driven oncology. Nature reviews Genetics. 2019;20(2):71–88. [PubMed: 30410101]
- 101.
- Lissa D, Robles AI. Methylation analyses in liquid biopsy. Translational lung cancer research. 2016;5(5):492–504. [PMC free article: PMC5099516] [PubMed: 27826530]
- 102.
- Bocchini M, Nicolini F, Severi S, Bongiovanni A, Ibrahim T, Simonetti G, et al. Biomarkers for Pancreatic Neuroendocrine Neoplasms (PanNENs) Management-An Updated Review. Frontiers in oncology. 2020;10:831. [PMC free article: PMC7267066] [PubMed: 32537434]
- 103.
- Öberg K, Califano A, Strosberg JR, Ma S, Pape U, Bodei L, et al. A meta-analysis of the accuracy of a neuroendocrine tumor mRNA genomic biomarker (NETest) in blood. Annals of oncology : official journal of the European Society for Medical Oncology. 2020;31(2):202–12. [PubMed: 31959337]
- 104.
- Oberg K, Modlin IM, De Herder W, Pavel M, Klimstra D, Frilling A, et al. Consensus on biomarkers for neuroendocrine tumour disease. The Lancet Oncology. 2015;16(9):e435–e46. [PMC free article: PMC5023063] [PubMed: 26370353]
- 105.
- Parbhu SK, Adler DG. Pancreatic neuroendocrine tumors: contemporary diagnosis and management. Hospital practice (1995). 2016;44(3):109-19. [PubMed: 27404266]
- 106.
- de Herder WW. Biochemistry of neuroendocrine tumours. Best practice & research Clinical endocrinology & metabolism. 2007;21(1):33–41. [PubMed: 17382264]
- 107.
- Ardill JE. Circulating markers for endocrine tumours of the gastroenteropancreatic tract. Annals of clinical biochemistry. 2008;45(Pt 6):539–59. [PubMed: 18941127]
- 108.
- Oberg K. Biochemical diagnosis of neuroendocrine GEP tumor. The Yale journal of biology and medicine. 1997;70(5-6):501–8. [PMC free article: PMC2589273] [PubMed: 9825477]
- 109.
- Panzuto F, Severi C, Cannizzaro R, Falconi M, Angeletti S, Pasquali A, et al. Utility of combined use of plasma levels of chromogranin A and pancreatic polypeptide in the diagnosis of gastrointestinal and pancreatic endocrine tumors. Journal of endocrinological investigation. 2004;27(1):6–11. [PubMed: 15053236]
- 110.
- Bajetta E, Ferrari L, Martinetti A, Celio L, Procopio G, Artale S, et al. Chromogranin A, neuron specific enolase, carcinoembryonic antigen, and hydroxyindole acetic acid evaluation in patients with neuroendocrine tumors. Cancer. 1999;86(5):858–65. [PubMed: 10463986]
- 111.
- Stabile BE, Howard TJ, Passaro E, Jr., O'Connor DT. Source of plasma chromogranin A elevation in gastrinoma patients. Archives of surgery (Chicago, Ill : 1960). 1990;125(4):451-3. [PubMed: 2322109]
- 112.
- Qiao XW, Qiu L, Chen YJ, Meng CT, Sun Z, Bai CM, et al. Chromogranin A is a reliable serum diagnostic biomarker for pancreatic neuroendocrine tumors but not for insulinomas. BMC endocrine disorders. 2014;14:64. [PMC free article: PMC4130880] [PubMed: 25099181]
- 113.
- Nobels FR, Kwekkeboom DJ, Coopmans W, Schoenmakers CH, Lindemans J, De Herder WW, et al. Chromogranin A as serum marker for neuroendocrine neoplasia: comparison with neuron-specific enolase and the alpha-subunit of glycoprotein hormones. The Journal of clinical endocrinology and metabolism. 1997;82(8):2622–8. [PubMed: 9253344]
- 114.
- Bílek R, Šafařík L, Ciprová V, Vlcek P, Lisá L. Chromogranin A, a member of neuroendocrine secretory proteins as a selective marker for laboratory diagnosis of pheochromocytoma. Physiological research. 2008;57 Suppl 1:S171–s9. [PubMed: 18271679]
- 115.
- Marotta V, Zatelli MC, Sciammarella C, Ambrosio MR, Bondanelli M, Colao A, et al. Chromogranin A as circulating marker for diagnosis and management of neuroendocrine neoplasms: more flaws than fame. Endocrine-related cancer. 2018;25(1):R11–r29. [PubMed: 29066503]
- 116.
- Stridsberg M, Eriksson B, Fellström B, Kristiansson G, Tiensuu Janson E. Measurements of chromogranin B can serve as a complement to chromogranin A. Regulatory peptides. 2007;139(1-3):80–3. [PubMed: 17116339]
- 117.
- Takiyyuddin MA, Cervenka JH, Hsiao RJ, Barbosa JA, Parmer RJ, O'Connor DT. Chromogranin A. Storage and release in hypertension. Hypertension (Dallas, Tex : 1979). 1990;15(3):237-46. [PubMed: 2406199]
- 118.
- Lindholm DP, Oberg K. Biomarkers and molecular imaging in gastroenteropancreatic neuroendocrine tumors. Hormone and metabolic research = Hormon- und Stoffwechselforschung = Hormones et metabolisme. 2011;43(12):832-7. [PubMed: 22009449]
- 119.
- Jilesen AP, Busch OR, van Gulik TM, Gouma DJ, Nieveen van Dijkum EJ. Standard pre- and postoperative determination of chromogranin a in resectable non-functioning pancreatic neuroendocrine tumors--diagnostic accuracy: NF-pNET and low tumor burden. Digestive surgery. 2014;31(6):407–14. [PubMed: 25572908]
- 120.
- Monaghan PJ, Lamarca A, Valle JW, Hubner RA, Mansoor W, Trainer PJ, et al. Routine measurement of plasma chromogranin B has limited clinical utility in the management of patients with neuroendocrine tumours. Clinical endocrinology. 2016;84(3):348–52. [PubMed: 26608723]
- 121.
- Hofland J, Zandee WT, de Herder WW. Role of biomarker tests for diagnosis of neuroendocrine tumours. Nature reviews Endocrinology. 2018;14(11):656–69. [PubMed: 30158549]
- 122.
- van Adrichem RC, Kamp K, Vandamme T, Peeters M, Feelders RA, de Herder WW. Serum neuron-specific enolase level is an independent predictor of overall survival in patients with gastroenteropancreatic neuroendocrine tumors. Annals of oncology : official journal of the European Society for Medical Oncology. 2016;27(4):746–7. [PubMed: 26712902]
- 123.
- Stronge RL, Turner GB, Johnston BT, McCance DR, McGinty A, Patterson CC, et al. A rapid rise in circulating pancreastatin in response to somatostatin analogue therapy is associated with poor survival in patients with neuroendocrine tumours. Annals of clinical biochemistry. 2008;45(Pt 6):560–6. [PubMed: 18782815]
- 124.
- Walter T, Chardon L, Chopin-laly X, Raverot V, Caffin AG, Chayvialle JA, et al. Is the combination of chromogranin A and pancreatic polypeptide serum determinations of interest in the diagnosis and follow-up of gastro-entero-pancreatic neuroendocrine tumours? European journal of cancer (Oxford, England : 1990). 2012;48(12):1766-73. [PubMed: 22133573]
- 125.
- Brown JE, Cook RJ, Major P, Lipton A, Saad F, Smith M, et al. Bone turnover markers as predictors of skeletal complications in prostate cancer, lung cancer, and other solid tumors. Journal of the National Cancer Institute. 2005;97(1):59–69. [PubMed: 15632381]
- 126.
- Lipton A, Costa L, Ali S, Demers L. Use of markers of bone turnover for monitoring bone metastases and the response to therapy. Seminars in oncology. 2001;28(4) Suppl 11:54–9. [PubMed: 11544577]
- 127.
- Khan MS, Kirkwood A, Tsigani T, Garcia-Hernandez J, Hartley JA, Caplin ME, et al. Circulating tumor cells as prognostic markers in neuroendocrine tumors. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2013;31(3):365–72. [PubMed: 23248251]
- 128.
- Mandair D, Khan MS, Lopes A, Furtado O'Mahony L, Ensell L, Lowe H, et al. Prognostic Threshold for Circulating Tumor Cells in Patients With Pancreatic and Midgut Neuroendocrine Tumors. The Journal of clinical endocrinology and metabolism. 2021;106(3):872–82. [PubMed: 33180939]
- 129.
- Khan MS, Tsigani T, Rashid M, Rabouhans JS, Yu D, Luong TV, et al. Circulating tumor cells and EpCAM expression in neuroendocrine tumors. Clinical cancer research : an official journal of the American Association for Cancer Research. 2011;17(2):337–45. [PubMed: 21224371]
- 130.
- Rizzo FM, Vesely C, Childs A, Marafioti T, Khan MS, Mandair D, et al. Circulating tumour cells and their association with bone metastases in patients with neuroendocrine tumours. British journal of cancer. 2019;120(3):294–300. [PMC free article: PMC6353867] [PubMed: 30636773]
- 131.
- Grande E, Capdevila J, Castellano D, Teulé A, Durán I, Fuster J, et al. Pazopanib in pretreated advanced neuroendocrine tumors: a phase II, open-label trial of the Spanish Task Force Group for Neuroendocrine Tumors (GETNE). Annals of oncology : official journal of the European Society for Medical Oncology. 2015;26(9):1987–93. [PubMed: 26063633]
- 132.
- Meyer T, Caplin M, Khan MS, Toumpanakis C, Shetty S, Ramage JK, et al. Circulating tumor cells and tumor biomarkers in functional midgut neuroendocrine tumors. Journal of Neuroendocrinology. 2022 [PMC free article: PMC9285714] [PubMed: 35132704]
- 133.
- Childs A, Vesely C, Ensell L, Lowe H, Luong TV, Caplin ME, et al. Expression of somatostatin receptors 2 and 5 in circulating tumour cells from patients with neuroendocrine tumours. British journal of cancer. 2016;115(12):1540–7. [PMC free article: PMC5155369] [PubMed: 27875519]
- 134.
- Rizzo FM, Meyer T. Liquid Biopsies for Neuroendocrine Tumors: Circulating Tumor Cells, DNA, and MicroRNAs. Endocrinology and metabolism clinics of North America. 2018;47(3):471–83. [PubMed: 30098711]
- 135.
- Mader S, Pantel K. Liquid Biopsy: Current Status and Future Perspectives. Oncology research and treatment. 2017;40(7-8):404–8. [PubMed: 28693023]
- 136.
- Boons G, Vandamme T, Peeters M, Beyens M, Driessen A, Janssens K, et al. Cell-Free DNA From Metastatic Pancreatic Neuroendocrine Tumor Patients Contains Tumor-Specific Mutations and Copy Number Variations. Frontiers in oncology. 2018;8:467. [PMC free article: PMC6221938] [PubMed: 30443491]
- 137.
- Malczewska A, Kidd M, Matar S, Kos-Kudla B, Modlin IM. A Comprehensive Assessment of the Role of miRNAs as Biomarkers in Gastroenteropancreatic Neuroendocrine Tumors. Neuroendocrinology. 2018;107(1):73–90. [PubMed: 29566385]
- 138.
- Li SC, Khan M, Caplin M, Meyer T, Öberg K, Giandomenico V. Somatostatin Analogs Treated Small Intestinal Neuroendocrine Tumor Patients Circulating MicroRNAs. PloS one. 2015;10(5):e0125553. [PMC free article: PMC4420277] [PubMed: 25942502]
- 139.
- Kövesdi A, Kurucz PA, Nyírő G, Darvasi O, Patócs A, Butz H. Circulating miRNA Increases the Diagnostic Accuracy of Chromogranin A in Metastatic Pancreatic Neuroendocrine Tumors. Cancers. 2020;12(9) [PMC free article: PMC7565801] [PubMed: 32887459]
- 140.
- Malczewska A, Kos-Kudła B, Kidd M, Drozdov I, Bodei L, Matar S, et al. The clinical applications of a multigene liquid biopsy (NETest) in neuroendocrine tumors. Advances in medical sciences. 2020;65(1):18–29. [PMC free article: PMC7453408] [PubMed: 31841822]
- 141.
- Modlin IM, Drozdov I, Kidd M. The identification of gut neuroendocrine tumor disease by multiple synchronous transcript analysis in blood. PloS one. 2013;8(5):e63364. [PMC free article: PMC3655166] [PubMed: 23691035]
- 142.
- Modlin IM, Kidd M, Falconi M, Filosso PL, Frilling A, Malczewska A, et al. A multigenomic liquid biopsy biomarker for neuroendocrine tumor disease outperforms CgA and has surgical and clinical utility. Annals of oncology : official journal of the European Society for Medical Oncology. 2021;32(11):1425–33. [PMC free article: PMC9393904] [PubMed: 34390828]
- 143.
- Modlin IM, Kidd M, Malczewska A, Drozdov I, Bodei L, Matar S, et al. The NETest: The Clinical Utility of Multigene Blood Analysis in the Diagnosis and Management of Neuroendocrine Tumors. Endocrinology and metabolism clinics of North America. 2018;47(3):485–504. [PMC free article: PMC6716518] [PubMed: 30098712]
- 144.
- Modlin IM, Frilling A, Salem RR, Alaimo D, Drymousis P, Wasan HS, et al. Blood measurement of neuroendocrine gene transcripts defines the effectiveness of operative resection and ablation strategies. Surgery. 2016;159(1):336–47. [PubMed: 26456125]
- 145.
- Ćwikła JB, Bodei L, Kolasinska-Ćwikła A, Sankowski A, Modlin IM, Kidd M. Circulating Transcript Analysis (NETest) in GEP-NETs Treated With Somatostatin Analogs Defines Therapy. The Journal of clinical endocrinology and metabolism. 2015;100(11):E1437–45. [PubMed: 26348352]
- 146.
- Bodei L, Kidd M, Modlin IM, Severi S, Drozdov I, Nicolini S, et al. Measurement of circulating transcripts and gene cluster analysis predicts and defines therapeutic efficacy of peptide receptor radionuclide therapy (PRRT) in neuroendocrine tumors. European journal of nuclear medicine and molecular imaging. 2016;43(5):839–51. [PubMed: 26596723]
- 147.
- van Treijen MJC, Korse CM, van Leeuwaarde RS, Saveur LJ, Vriens MR, Verbeek WHM, et al. Blood Transcript Profiling for the Detection of Neuroendocrine Tumors: Results of a Large Independent Validation Study. Frontiers in endocrinology. 2018;9:740. [PMC free article: PMC6288275] [PubMed: 30564197]
- 148.
- Lee L, Ito T, Jensen RT. Imaging of pancreatic neuroendocrine tumors: recent advances, current status, and controversies. Expert review of anticancer therapy. 2018;18(9):837–60. [PMC free article: PMC6283410] [PubMed: 29973077]
- 149.
- Akirov A, Larouche V, Alshehri S, Asa SL, Ezzat S. Treatment Options for Pancreatic Neuroendocrine Tumors. Cancers. 2019;11(6) [PMC free article: PMC6628351] [PubMed: 31207914]
- 150.
- Belotto M, Crouzillard B, Araujo KO, Peixoto RD. PANCREATIC NEUROENDOCRINE TUMORS: SURGICAL RESECTION. Arquivos brasileiros de cirurgia digestiva : ABCD = Brazilian archives of digestive surgery. 2019;32(1):e1428. [PMC free article: PMC6368164] [PubMed: 30758476]
- 151.
- Zerbi A, Capitanio V, Boninsegna L, Pasquali C, Rindi G, Delle Fave G, et al. Surgical treatment of pancreatic endocrine tumours in Italy: results of a prospective multicentre study of 262 cases. Langenbeck's archives of surgery. 2011;396(3):313–21. [PubMed: 20857140]
- 152.
- Hill JS, McPhee JT, McDade TP, Zhou Z, Sullivan ME, Whalen GF, et al. Pancreatic neuroendocrine tumors: the impact of surgical resection on survival. Cancer. 2009;115(4):741–51. [PubMed: 19130464]
- 153.
- Gomez-Rivera F, Stewart AE, Arnoletti JP, Vickers S, Bland KI, Heslin MJ. Surgical treatment of pancreatic endocrine neoplasms. American journal of surgery. 2007;193(4):460–5. [PubMed: 17368289]
- 154.
- Martin RC, Kooby DA, Weber SM, Merchant NB, Parikh AA, Cho CS, et al. Analysis of 6,747 pancreatic neuroendocrine tumors for a proposed staging system. Journal of gastrointestinal surgery : official journal of the Society for Surgery of the Alimentary Tract. 2011;15(1):175–83. [PubMed: 21103949]
- 155.
- Scarpa A, Mantovani W, Capelli P, Beghelli S, Boninsegna L, Bettini R, et al. Pancreatic endocrine tumors: improved TNM staging and histopathological grading permit a clinically efficient prognostic stratification of patients. Modern pathology : an official journal of the United States and Canadian Academy of Pathology. Inc. 2010;23(6):824–33. [PubMed: 20305616]
- 156.
- Toste PA, Kadera BE, Tatishchev SF, Dawson DW, Clerkin BM, Muthusamy R, et al. Nonfunctional pancreatic neuroendocrine tumors <2 cm on preoperative imaging are associated with a low incidence of nodal metastasis and an excellent overall survival. Journal of gastrointestinal surgery : official journal of the Society for Surgery of the Alimentary Tract. 2013;17(12):2105–13. [PubMed: 24101447]
- 157.
- Partelli S, Gaujoux S, Boninsegna L, Cherif R, Crippa S, Couvelard A, et al. Pattern and clinical predictors of lymph node involvement in nonfunctioning pancreatic neuroendocrine tumors (NF-PanNETs). JAMA surgery. 2013;148(10):932–9. [PubMed: 23986355]
- 158.
- Titan AL, Norton JA, Fisher AT, Foster DS, Harris EJ, Worhunsky DJ, et al. Evaluation of Outcomes Following Surgery for Locally Advanced Pancreatic Neuroendocrine Tumors. JAMA network open. 2020;3(11):e2024318. [PMC free article: PMC7643030] [PubMed: 33146734]
- 159.
- Bettini R, Partelli S, Boninsegna L, Capelli P, Crippa S, Pederzoli P, et al. Tumor size correlates with malignancy in nonfunctioning pancreatic endocrine tumor. Surgery. 2011;150(1):75–82. [PubMed: 21683859]
- 160.
- Gaujoux S, Partelli S, Maire F, D'Onofrio M, Larroque B, Tamburrino D, et al. Observational study of natural history of small sporadic nonfunctioning pancreatic neuroendocrine tumors. The Journal of clinical endocrinology and metabolism. 2013;98(12):4784–9. [PubMed: 24057286]
- 161.
- Triponez F, Sadowski SM, Pattou F, Cardot-Bauters C, Mirallié E, Le Bras M, et al. Long-term Follow-up of MEN1 Patients Who Do Not Have Initial Surgery for Small ≤2 cm Nonfunctioning Pancreatic Neuroendocrine Tumors, an AFCE and GTE Study: Association Francophone de Chirurgie Endocrinienne & Groupe d'Etude des Tumeurs Endocrines. Annals of surgery. 2018;268(1):158–64. [PMC free article: PMC6012055] [PubMed: 28263205]
- 162.
- Thakker RV, Newey PJ, Walls GV, Bilezikian J, Dralle H, Ebeling PR, et al. Clinical practice guidelines for multiple endocrine neoplasia type 1 (MEN1). The Journal of clinical endocrinology and metabolism. 2012;97(9):2990–3011. [PubMed: 22723327]
- 163.
- Krug S, Gress TM, Michl P, Rinke A. The Role of Cytotoxic Chemotherapy in Advanced Pancreatic Neuroendocrine Tumors. Digestion. 2017;96(2):67–75. [PubMed: 28728148]
- 164.
- Akerström G, Hellman P. Surgery on neuroendocrine tumours. Best practice & research Clinical endocrinology & metabolism. 2007;21(1):87–109. [PubMed: 17382267]
- 165.
- Jensen RT, Norton JA. Treatment of Pancreatic Neuroendocrine Tumors in Multiple Endocrine Neoplasia Type 1: Some Clarity But Continued Controversy. Pancreas. 2017;46(5):589–94. [PMC free article: PMC5407310] [PubMed: 28426491]
- 166.
- Jensen RT, Niederle B, Mitry E, Ramage JK, Steinmuller T, Lewington V, et al. Gastrinoma (duodenal and pancreatic). Neuroendocrinology. 2006;84(3):173–82. [PubMed: 17312377]
- 167.
- Norton JA, Fraker DL, Alexander HR, Venzon DJ, Doppman JL, Serrano J, et al. Surgery to cure the Zollinger-Ellison syndrome. The New England journal of medicine. 1999;341(9):635–44. [PubMed: 10460814]
- 168.
- Al-Kurd A, Chapchay K, Grozinsky-Glasberg S, Mazeh H. Laparoscopic resection of pancreatic neuroendocrine tumors. World journal of gastroenterology. 2014;20(17):4908–16. [PMC free article: PMC4009522] [PubMed: 24803802]
- 169.
- Drymousis P, Raptis DA, Spalding D, Fernandez-Cruz L, Menon D, Breitenstein S, et al. Laparoscopic versus open pancreas resection for pancreatic neuroendocrine tumours: a systematic review and meta-analysis. HPB : the official journal of the International Hepato Pancreato Biliary Association. 2014;16(5):397–406. [PMC free article: PMC4008158] [PubMed: 24245906]
- 170.
- Venkat R, Edil BH, Schulick RD, Lidor AO, Makary MA, Wolfgang CL. Laparoscopic distal pancreatectomy is associated with significantly less overall morbidity compared to the open technique: a systematic review and meta-analysis. Annals of surgery. 2012;255(6):1048–59. [PubMed: 22511003]
- 171.
- Kendrick ML, van Hilst J, Boggi U, de Rooij T, Walsh RM, Zeh HJ, et al. Minimally invasive pancreatoduodenectomy. HPB : the official journal of the International Hepato Pancreato Biliary Association. 2017;19(3):215–24. [PubMed: 28317658]
- 172.
- Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer. 2003;97(4):934–59. [PubMed: 12569593]
- 173.
- Chamberlain RS, Canes D, Brown KT, Saltz L, Jarnagin W, Fong Y, et al. Hepatic neuroendocrine metastases: does intervention alter outcomes? Journal of the American College of Surgeons. 2000;190(4):432–45. [PubMed: 10757381]
- 174.
- Hodul PJ, Strosberg JR, Kvols LK. Aggressive surgical resection in the management of pancreatic neuroendocrine tumors: when is it indicated? Cancer control : journal of the Moffitt Cancer Center. 2008;15(4):314-21. [PubMed: 18813199]
- 175.
- Zhou B, Zhan C, Ding Y, Yan S, Zheng S. Role of palliative resection of the primary pancreatic neuroendocrine tumor in patients with unresectable metastatic liver disease: a systematic review and meta-analysis. OncoTargets and therapy. 2018;11:975–82. [PMC free article: PMC5827741] [PubMed: 29503572]
- 176.
- Mazzaferro V, Pulvirenti A, Coppa J. Neuroendocrine tumors metastatic to the liver: how to select patients for liver transplantation? Journal of hepatology. 2007;47(4):460–6. [PubMed: 17697723]
- 177.
- Shah MH, Goldner WS, Halfdanarson TR, Bergsland E, Berlin JD, Halperin D, et al. NCCN Guidelines Insights: Neuroendocrine and Adrenal Tumors, Version 2.2018. Journal of the National Comprehensive Cancer Network : JNCCN. 2018;16(6):693–702. [PubMed: 29891520]
- 178.
- Solorzano CC, Lee JE, Pisters PW, Vauthey JN, Ayers GD, Jean ME, et al. Nonfunctioning islet cell carcinoma of the pancreas: survival results in a contemporary series of 163 patients. Surgery. 2001;130(6):1078–85. [PubMed: 11742342]
- 179.
- Elias D, Lefevre JH, Duvillard P, Goéré D, Dromain C, Dumont F, et al. Hepatic metastases from neuroendocrine tumors with a "thin slice" pathological examination: they are many more than you think. Annals of surgery. 2010;251(2):307–10. [PubMed: 20010089]
- 180.
- Sarmiento JM, Heywood G, Rubin J, Ilstrup DM, Nagorney DM, Que FG. Surgical treatment of neuroendocrine metastases to the liver: a plea for resection to increase survival. Journal of the American College of Surgeons. 2003;197(1):29–37. [PubMed: 12831921]
- 181.
- Norton JA, Sugarbaker PH, Doppman JL, Wesley RA, Maton PN, Gardner JD, et al. Aggressive resection of metastatic disease in selected patients with malignant gastrinoma. Annals of surgery. 1986;203(4):352–9. [PMC free article: PMC1251117] [PubMed: 2938550]
- 182.
- Sarmiento JM, Que FG. Hepatic surgery for metastases from neuroendocrine tumors. Surgical oncology clinics of North America. 2003;12(1):231–42. [PubMed: 12735141]
- 183.
- House MG, Cameron JL, Lillemoe KD, Schulick RD, Choti MA, Hansel DE, et al. Differences in survival for patients with resectable versus unresectable metastases from pancreatic islet cell cancer. Journal of gastrointestinal surgery : official journal of the Society for Surgery of the Alimentary Tract. 2006;10(1):138–45. [PubMed: 16368504]
- 184.
- Gupta S, Johnson MM, Murthy R, Ahrar K, Wallace MJ, Madoff DC, et al. Hepatic arterial embolization and chemoembolization for the treatment of patients with metastatic neuroendocrine tumors: variables affecting response rates and survival. Cancer. 2005;104(8):1590–602. [PubMed: 16134179]
- 185.
- Mazzaferro V, Sposito C, Coppa J, Miceli R, Bhoori S, Bongini M, et al. The Long-Term Benefit of Liver Transplantation for Hepatic Metastases From Neuroendocrine Tumors. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons. 2016;16(10):2892–902. [PubMed: 27134017]
- 186.
- Herrera-Martínez AD, Hofland J, Hofland LJ, Brabander T, Eskens F, Gálvez Moreno MA, et al. Targeted Systemic Treatment of Neuroendocrine Tumors: Current Options and Future Perspectives. Drugs. 2019;79(1):21–42. [PMC free article: PMC6338796] [PubMed: 30560479]
- 187.
- Gomes-Porras M, Cárdenas-Salas J, Álvarez-Escolá C. Somatostatin Analogs in Clinical Practice: a Review. International journal of molecular sciences. 2020;21(5) [PMC free article: PMC7084228] [PubMed: 32121432]
- 188.
- Hauser H, Gerson DS, Reidy-Lagunes D, Raj N. Systemic Therapies for Metastatic Pancreatic Neuroendocrine Tumors. Current treatment options in oncology. 2019;20(12):87. [PubMed: 31776785]
- 189.
- Caplin ME, Pavel M, Ćwikła JB, Phan AT, Raderer M, Sedláčková E, et al. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. The New England journal of medicine. 2014;371(3):224–33. [PubMed: 25014687]
- 190.
- Caplin ME, Pavel M, Ćwikła JB, Phan AT, Raderer M, Sedláčková E, et al. Anti-tumour effects of lanreotide for pancreatic and intestinal neuroendocrine tumours: the CLARINET open-label extension study. Endocrine-related cancer. 2016;23(3):191–9. [PMC free article: PMC4740728] [PubMed: 26743120]
- 191.
- Pavel M, Öberg K, Falconi M, Krenning EP, Sundin A, Perren A, et al. Gastroenteropancreatic neuroendocrine neoplasms: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of oncology : official journal of the European Society for Medical Oncology. 2020;31(7):844–60. [PubMed: 32272208]
- 192.
- Vinik AI, Raymond E. Pancreatic neuroendocrine tumors: approach to treatment with focus on sunitinib. Therapeutic advances in gastroenterology. 2013;6(5):396–411. [PMC free article: PMC3756637] [PubMed: 24003340]
- 193.
- Kulke MH, Lenz HJ, Meropol NJ, Posey J, Ryan DP, Picus J, et al. Activity of sunitinib in patients with advanced neuroendocrine tumors. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2008;26(20):3403–10. [PubMed: 18612155]
- 194.
- Raymond E, Dahan L, Raoul JL, Bang YJ, Borbath I, Lombard-Bohas C, et al. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. The New England journal of medicine. 2011;364(6):501–13. [PubMed: 21306237]
- 195.
- Faivre S, Niccoli P, Castellano D, Valle JW, Hammel P, Raoul JL, et al. Sunitinib in pancreatic neuroendocrine tumors: updated progression-free survival and final overall survival from a phase III randomized study. Annals of oncology : official journal of the European Society for Medical Oncology. 2017;28(2):339–43. [PubMed: 27836885]
- 196.
- Vinik E, Carlton CA, Silva MP, Vinik AI. Development of the Norfolk quality of life tool for assessing patients with neuroendocrine tumors. Pancreas. 2009;38(3):e87–95. [PubMed: 19276865]
- 197.
- Yao JC, Lombard-Bohas C, Baudin E, Kvols LK, Rougier P, Ruszniewski P, et al. Daily oral everolimus activity in patients with metastatic pancreatic neuroendocrine tumors after failure of cytotoxic chemotherapy: a phase II trial. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2010;28(1):69–76. [PMC free article: PMC4295034] [PubMed: 19933912]
- 198.
- Yao JC, Shah MH, Ito T, Bohas CL, Wolin EM, Van Cutsem E, et al. Everolimus for advanced pancreatic neuroendocrine tumors. The New England journal of medicine. 2011;364(6):514–23. [PMC free article: PMC4208619] [PubMed: 21306238]
- 199.
- Lombard-Bohas C, Yao JC, Hobday T, Van Cutsem E, Wolin EM, Panneerselvam A, et al. Impact of prior chemotherapy use on the efficacy of everolimus in patients with advanced pancreatic neuroendocrine tumors: a subgroup analysis of the phase III RADIANT-3 trial. Pancreas. 2015;44(2):181–9. [PMC free article: PMC4327560] [PubMed: 25479584]
- 200.
- Chan JA, Blaszkowsky L, Stuart K, Zhu AX, Allen J, Wadlow R, et al. A prospective, phase 1/2 study of everolimus and temozolomide in patients with advanced pancreatic neuroendocrine tumor. Cancer. 2013;119(17):3212–8. [PMC free article: PMC4308727] [PubMed: 23733618]
- 201.
- Kulke MH, Ruszniewski P, Van Cutsem E, Lombard-Bohas C, Valle JW, De Herder WW, et al. A randomized, open-label, phase 2 study of everolimus in combination with pasireotide LAR or everolimus alone in advanced, well-differentiated, progressive pancreatic neuroendocrine tumors: COOPERATE-2 trial. Annals of oncology : official journal of the European Society for Medical Oncology. 2017;28(6):1309–15. [PMC free article: PMC7360140] [PubMed: 28327907]
- 202.
- Halfdanarson TR, Strosberg JR, Tang L, Bellizzi AM, Bergsland EK, O'Dorisio TM, et al. The North American Neuroendocrine Tumor Society Consensus Guidelines for Surveillance and Medical Management of Pancreatic Neuroendocrine Tumors. Pancreas. 2020;49(7):863–81. [PubMed: 32675783]
- 203.
- Yao J, Phan AT. Optimising Therapeutic Options for Patients with Advanced Pancreatic Neuroendocrine Tumours. European Oncology & Haematology. 2012;8(4):217–23.
- 204.
- Mateo J, Heymach JV, Zurita AJ. Circulating biomarkers of response to sunitinib in gastroenteropancreatic neuroendocrine tumors: current data and clinical outlook. Molecular diagnosis & therapy. 2012;16(3):151–61. [PMC free article: PMC3872147] [PubMed: 22515658]
- 205.
- Bello C, Deprimo SE, Friece C, Smeraglia J, Sherman L, Tye L, et al. Analysis of circulating biomarkers of sunitinib malate in patients with unresectable neuroendocrine tumors (NET): VEGF, IL-8, and soluble VEGF receptors 2 and 3. Journal of Clinical Oncology. 2006;24(18) suppl:4045.
- 206.
- Yao JC, Pavel M, Phan AT, Kulke MH, Hoosen S, St Peter J, et al. Chromogranin A and neuron-specific enolase as prognostic markers in patients with advanced pNET treated with everolimus. The Journal of clinical endocrinology and metabolism. 2011;96(12):3741–9. [PubMed: 21994954]
- 207.
- Palmieri LJ, Dermine S, Barré A, Dhooge M, Brezault C, Cottereau AS, et al. Medical Treatment of Advanced Pancreatic Neuroendocrine Neoplasms. Journal of clinical medicine. 2020;9(6) [PMC free article: PMC7355438] [PubMed: 32549203]
- 208.
- Pavel M, O'Toole D, Costa F, Capdevila J, Gross D, Kianmanesh R, et al. ENETS Consensus Guidelines Update for the Management of Distant Metastatic Disease of Intestinal, Pancreatic, Bronchial Neuroendocrine Neoplasms (NEN) and NEN of Unknown Primary Site. Neuroendocrinology. 2016;103(2):172–85. [PubMed: 26731013]
- 209.
- Okusaka T, Ueno H, Morizane C, Kondo S, Sakamoto Y, Takahashi H, et al. Cytotoxic chemotherapy for pancreatic neuroendocrine tumors. Journal of hepato-biliary-pancreatic sciences. 2015;22(8):628–33. [PubMed: 25940377]
- 210.
- Moertel CG, Lefkopoulo M, Lipsitz S, Hahn RG, Klaassen D. Streptozocin-doxorubicin, streptozocin-fluorouracil or chlorozotocin in the treatment of advanced islet-cell carcinoma. The New England journal of medicine. 1992;326(8):519–23. [PubMed: 1310159]
- 211.
- Kouvaraki MA, Ajani JA, Hoff P, Wolff R, Evans DB, Lozano R, et al. Fluorouracil, doxorubicin, and streptozocin in the treatment of patients with locally advanced and metastatic pancreatic endocrine carcinomas. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2004;22(23):4762–71. [PubMed: 15570077]
- 212.
- Ramanathan RK, Cnaan A, Hahn RG, Carbone PP, Haller DG. Phase II trial of dacarbazine (DTIC) in advanced pancreatic islet cell carcinoma. Study of the Eastern Cooperative Oncology Group-E6282. Annals of oncology : official journal of the European Society for Medical Oncology. 2001;12(8):1139–43. [PubMed: 11583197]
- 213.
- de Mestier L, Walter T, Brixi H, Evrard C, Legoux JL, de Boissieu P, et al. Comparison of Temozolomide-Capecitabine to 5-Fluorouracile-Dacarbazine in 247 Patients with Advanced Digestive Neuroendocrine Tumors Using Propensity Score Analyses. Neuroendocrinology. 2019;108(4):343–53. [PubMed: 30759445]
- 214.
- Strosberg JR, Fine RL, Choi J, Nasir A, Coppola D, Chen DT, et al. First-line chemotherapy with capecitabine and temozolomide in patients with metastatic pancreatic endocrine carcinomas. Cancer. 2011;117(2):268–75. [PMC free article: PMC4665634] [PubMed: 20824724]
- 215.
- Chan JA, Stuart K, Earle CC, Clark JW, Bhargava P, Miksad R, et al. Prospective study of bevacizumab plus temozolomide in patients with advanced neuroendocrine tumors. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2012;30(24):2963–8. [PMC free article: PMC4874232] [PubMed: 22778320]
- 216.
- Koumarianou A, Antoniou S, Kanakis G, Economopoulos N, Rontogianni D, Ntavatzikos A, et al. Combination treatment with metronomic temozolomide, bevacizumab and long-acting octreotide for malignant neuroendocrine tumours. Endocrine-related cancer. 2012;19(1):L1–4. [PMC free article: PMC3255566] [PubMed: 22232542]
- 217.
- Kulke MH, Stuart K, Enzinger PC, Ryan DP, Clark JW, Muzikansky A, et al. Phase II study of temozolomide and thalidomide in patients with metastatic neuroendocrine tumors. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2006;24(3):401–6. [PubMed: 16421420]
- 218.
- Kunz PL, Catalano PJ, Nimeiri H, Fisher GA, Longacre TA, Suarez CJ, et al. A randomized study of temozolomide or temozolomide and capecitabine in patients with advanced pancreatic neuroendocrine tumors: A trial of the ECOG-ACRIN Cancer Research Group (E2211). Journal of Clinical Oncology. 2018;36(15) suppl:4004. [PMC free article: PMC9995105] [PubMed: 36260828]
- 219.
- de Mestier L, Walter T, Evrard C, de Boissieu P, Hentic O, Cros J, et al. Temozolomide Alone or Combined with Capecitabine for the Treatment of Advanced Pancreatic Neuroendocrine Tumor. Neuroendocrinology. 2020;110(1-2):83–91. [PMC free article: PMC6979423] [PubMed: 31071715]
- 220.
- Moertel CG, Kvols LK, O'Connell MJ, Rubin J. Treatment of neuroendocrine carcinomas with combined etoposide and cisplatin. Evidence of major therapeutic activity in the anaplastic variants of these neoplasms. Cancer. 1991;68(2):227–32. [PubMed: 1712661]
- 221.
- Mitry E, Baudin E, Ducreux M, Sabourin JC, Rufié P, Aparicio T, et al. Treatment of poorly differentiated neuroendocrine tumours with etoposide and cisplatin. British journal of cancer. 1999;81(8):1351–5. [PMC free article: PMC2362979] [PubMed: 10604732]
- 222.
- Thomas KEH, Voros BA, Boudreaux JP, Thiagarajan R, Woltering EA, Ramirez RA. Current Treatment Options in Gastroenteropancreatic Neuroendocrine Carcinoma. The oncologist. 2019;24(8):1076–88. [PMC free article: PMC6693730] [PubMed: 30635447]
- 223.
- Ilett EE, Langer SW, Olsen IH, Federspiel B, Kjær A, Knigge U. Neuroendocrine Carcinomas of the Gastroenteropancreatic System: A Comprehensive Review. Diagnostics (Basel, Switzerland). 2015;5(2):119–76. [PMC free article: PMC4665594] [PubMed: 26854147]
- 224.
- Sorbye H, Welin S, Langer SW, Vestermark LW, Holt N, Osterlund P, et al. Predictive and prognostic factors for treatment and survival in 305 patients with advanced gastrointestinal neuroendocrine carcinoma (WHO G3): the NORDIC NEC study. Annals of oncology : official journal of the European Society for Medical Oncology. 2013;24(1):152–60. [PubMed: 22967994]
- 225.
- Hentic O, Hammel P, Couvelard A, Rebours V, Zappa M, Palazzo M, et al. FOLFIRI regimen: an effective second-line chemotherapy after failure of etoposide-platinum combination in patients with neuroendocrine carcinomas grade 3. Endocrine-related cancer. 2012;19(6):751–7. [PubMed: 22940375]
- 226.
- Bajetta E, Catena L, Procopio G, De Dosso S, Bichisao E, Ferrari L, et al. Are capecitabine and oxaliplatin (XELOX) suitable treatments for progressing low-grade and high-grade neuroendocrine tumours? Cancer chemotherapy and pharmacology. 2007;59(5):637–42. [PubMed: 16937105]
- 227.
- Spada F, Antonuzzo L, Marconcini R, Radice D, Antonuzzo A, Ricci S, et al. Oxaliplatin-Based Chemotherapy in Advanced Neuroendocrine Tumors: Clinical Outcomes and Preliminary Correlation with Biological Factors. Neuroendocrinology. 2016;103(6):806–14. [PubMed: 26789262]
- 228.
- Zhu J, Strosberg JR, Dropkin E, Strickler JH. Treatment of High-Grade Metastatic Pancreatic Neuroendocrine Carcinoma with FOLFIRINOX. Journal of gastrointestinal cancer. 2015;46(2):166–9. [PubMed: 25662891]
- 229.
- Camus B, Cottereau AS, Palmieri LJ, Dermine S, Tenenbaum F, Brezault C, et al. Indications of Peptide Receptor Radionuclide Therapy (PRRT) in Gastroenteropancreatic and Pulmonary Neuroendocrine Tumors: An Updated Review. Journal of clinical medicine. 2021;10(6) [PMC free article: PMC8003169] [PubMed: 33803817]
- 230.
- Kwekkeboom DJ, Teunissen JJ, Bakker WH, Kooij PP, de Herder WW, Feelders RA, et al. Radiolabeled somatostatin analog [177Lu-DOTA0,Tyr3]octreotate in patients with endocrine gastroenteropancreatic tumors. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2005;23(12):2754–62. [PubMed: 15837990]
- 231.
- Vinik E, Silva MP, Vinik AI. Measuring the relationship of quality of life and health status, including tumor burden, symptoms, and biochemical measures in patients with neuroendocrine tumors. Endocrinology and metabolism clinics of North America. 2011;40(1):97–109. viii. [PubMed: 21349413]
- 232.
- Ezziddin S, Khalaf F, Vanezi M, Haslerud T, Mayer K, Al Zreiqat A, et al. Outcome of peptide receptor radionuclide therapy with 177Lu-octreotate in advanced grade 1/2 pancreatic neuroendocrine tumours. European journal of nuclear medicine and molecular imaging. 2014;41(5):925–33. [PubMed: 24504504]
- 233.
- Strosberg J, El-Haddad G, Wolin E, Hendifar A, Yao J, Chasen B, et al. Phase 3 Trial of (177)Lu-Dotatate for Midgut Neuroendocrine Tumors. The New England journal of medicine. 2017;376(2):125–35. [PMC free article: PMC5895095] [PubMed: 28076709]
- 234.
- Food and Drug Administration. FDA approves lutetium Lu 177 dotatate for treatment of GEP-NETS. 2018.
- 235.
- Bodei L, Mueller-Brand J, Baum RP, Pavel ME, Hörsch D, O'Dorisio MS, et al. The joint IAEA, EANM, and SNMMI practical guidance on peptide receptor radionuclide therapy (PRRNT) in neuroendocrine tumours. European journal of nuclear medicine and molecular imaging. 2013;40(5):800–16. [PMC free article: PMC3622744] [PubMed: 23389427]
- 236.
- Brabander T, van der Zwan WA, Teunissen JJM, Kam BLR, Feelders RA, de Herder WW, et al. Long-Term Efficacy, Survival, and Safety of [(177)Lu-DOTA(0),Tyr(3)]octreotate in Patients with Gastroenteropancreatic and Bronchial Neuroendocrine Tumors. Clinical cancer research : an official journal of the American Association for Cancer Research. 2017;23(16):4617–24. [PubMed: 28428192]
- 237.
- Satapathy S, Mittal BR. 177Lu-DOTATATE peptide receptor radionuclide therapy versus Everolimus in advanced pancreatic neuroendocrine tumors: a systematic review and meta-analysis. Nuclear medicine communications. 2019;40(12):1195–203. [PubMed: 31658219]
- 238.
- Pusceddu S, Prinzi N, Tafuto S, Ibrahim T, Filice A, Brizzi MP, et al. Association of Upfront Peptide Receptor Radionuclide Therapy With Progression-Free Survival Among Patients With Enteropancreatic Neuroendocrine Tumors. JAMA network open. 2022;5(2):e220290. [PMC free article: PMC8874344] [PubMed: 35201309]
- 239.
- Howe JR, Merchant NB, Conrad C, Keutgen XM, Hallet J, Drebin JA, et al. The North American Neuroendocrine Tumor Society Consensus Paper on the Surgical Management of Pancreatic Neuroendocrine Tumors. Pancreas. 2020;49(1):1–33. [PMC free article: PMC7029300] [PubMed: 31856076]
- 240.
- Bloomston M, Al-Saif O, Klemanski D, Pinzone JJ, Martin EW, Palmer B, et al. Hepatic artery chemoembolization in 122 patients with metastatic carcinoid tumor: lessons learned. Journal of gastrointestinal surgery : official journal of the Society for Surgery of the Alimentary Tract. 2007;11(3):264–71. [PubMed: 17458596]
- 241.
- Kennedy AS, Dezarn WA, McNeillie P, Coldwell D, Nutting C, Carter D, et al. Radioembolization for unresectable neuroendocrine hepatic metastases using resin 90Y-microspheres: early results in 148 patients. American journal of clinical oncology. 2008;31(3):271–9. [PubMed: 18525307]
- 242.
- Rhee TK, Lewandowski RJ, Liu DM, Mulcahy MF, Takahashi G, Hansen PD, et al. 90Y Radioembolization for metastatic neuroendocrine liver tumors: preliminary results from a multi-institutional experience. Annals of surgery. 2008;247(6):1029–35. [PubMed: 18520231]
- 243.
- Grozinsky-Glasberg S, Kaltsas G, Kaltsatou M, Lev-Cohain N, Klimov A, Vergadis V, et al. Hepatic intra-arterial therapies in metastatic neuroendocrine tumors: lessons from clinical practice. Endocrine. 2018;60(3):499–509. [PubMed: 29383678]
- 244.
- Patnaik A, Kang SP, Rasco D, Papadopoulos KP, Elassaiss-Schaap J, Beeram M, et al. Phase I Study of Pembrolizumab (MK-3475; Anti-PD-1 Monoclonal Antibody) in Patients with Advanced Solid Tumors. Clinical cancer research : an official journal of the American Association for Cancer Research. 2015;21(19):4286–93. [PubMed: 25977344]
- 245.
- McGranahan N, Furness AJ, Rosenthal R, Ramskov S, Lyngaa R, Saini SK, et al. Clonal neoantigens elicit T cell immunoreactivity and sensitivity to immune checkpoint blockade. Science (New York, NY). 2016;351(6280):1463–9. [PMC free article: PMC4984254] [PubMed: 26940869]
- 246.
- Mehnert JM, Bergsland E, O'Neil BH, Santoro A, Schellens JHM, Cohen RB, et al. Pembrolizumab for the treatment of programmed death-ligand 1-positive advanced carcinoid or pancreatic neuroendocrine tumors: Results from the KEYNOTE-028 study. Cancer. 2020;126(13):3021–30. [PubMed: 32320048]
- 247.
- Strosberg J, Mizuno N, Doi T, Grande E, Delord JP, Shapira-Frommer R, et al. Efficacy and Safety of Pembrolizumab in Previously Treated Advanced Neuroendocrine Tumors: Results From the Phase II KEYNOTE-158 Study. Clinical cancer research : an official journal of the American Association for Cancer Research. 2020;26(9):2124–30. [PMC free article: PMC7811789] [PubMed: 31980466]
- 248.
- Naing A, Papadopoulos KP, Autio KA, Ott PA, Patel MR, Wong DJ, et al. Safety, Antitumor Activity, and Immune Activation of Pegylated Recombinant Human Interleukin-10 (AM0010) in Patients With Advanced Solid Tumors. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2016;34(29):3562–9. [PMC free article: PMC5657013] [PubMed: 27528724]
- 249.
- Yao JC, Guthrie KA, Moran C, Strosberg JR, Kulke MH, Chan JA, et al. Phase III Prospective Randomized Comparison Trial of Depot Octreotide Plus Interferon Alfa-2b Versus Depot Octreotide Plus Bevacizumab in Patients With Advanced Carcinoid Tumors: SWOG S0518. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2017;35(15):1695–703. [PMC free article: PMC5455764] [PubMed: 28384065]
- 250.
- Ducreux M, Dahan L, Smith D, O'Toole D, Lepère C, Dromain C, et al. Bevacizumab combined with 5-FU/streptozocin in patients with progressive metastatic well-differentiated pancreatic endocrine tumours (BETTER trial)--a phase II non-randomised trial. European journal of cancer (Oxford, England : 1990). 2014;50(18):3098-106. [PubMed: 25454412]
- 251.
- Xu J, Shen L, Zhou Z, Li J, Bai C, Chi Y, et al. Surufatinib in advanced extrapancreatic neuroendocrine tumours (SANET-ep): a randomised, double-blind, placebo-controlled, phase 3 study. The Lancet Oncology. 2020;21(11):1500–12. [PubMed: 32966811]
- 252.
- Xu J, Shen L, Bai C, Wang W, Li J, Yu X, et al. Surufatinib in advanced pancreatic neuroendocrine tumours (SANET-p): a randomised, double-blind, placebo-controlled, phase 3 study. The Lancet Oncology. 2020;21(11):1489–99. [PubMed: 32966810]
- 253.
- Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. Journal of the National Cancer Institute. 1993;85(5):365–76. [PubMed: 8433390]
- 254.
- Yadegarfar G, Friend L, Jones L, Plum LM, Ardill J, Taal B, et al. Validation of the EORTC QLQ-GINET21 questionnaire for assessing quality of life of patients with gastrointestinal neuroendocrine tumours. British journal of cancer. 2013;108(2):301–10. [PMC free article: PMC3566824] [PubMed: 23322194]
- 255.
- Jiménez-Fonseca P, Carmona-Bayonas A, Martín-Pérez E, Crespo G, Serrano R, Llanos M, et al. Health-related quality of life in well-differentiated metastatic gastroenteropancreatic neuroendocrine tumors. Cancer metastasis reviews. 2015;34(3):381–400. [PubMed: 26245646]
- 256.
- Meng Y, McCarthy G, Berthon A, Dinet J. Patient-reported health state utilities in metastatic gastroenteropancreatic neuroendocrine tumours - an analysis based on the CLARINET study. Health and quality of life outcomes. 2017;15(1):131. [PMC free article: PMC5492941] [PubMed: 28662673]
- 257.
- Marinova M, Mücke M, Mahlberg L, Essler M, Cuhls H, Radbruch L, et al. Improving quality of life in patients with pancreatic neuroendocrine tumor following peptide receptor radionuclide therapy assessed by EORTC QLQ-C30. European journal of nuclear medicine and molecular imaging. 2018;45(1):38–46. [PubMed: 28864881]
- 258.
- Martini C, Gamper EM, Wintner L, Nilica B, Sperner-Unterweger B, Holzner B, et al. Systematic review reveals lack of quality in reporting health-related quality of life in patients with gastroenteropancreatic neuroendocrine tumours. Health and quality of life outcomes. 2016;14(1):127. [PMC free article: PMC5018190] [PubMed: 27614762]
- Review Models in Pancreatic Neuroendocrine Neoplasms: Current Perspectives and Future Directions.[Cancers (Basel). 2023]Review Models in Pancreatic Neuroendocrine Neoplasms: Current Perspectives and Future Directions.Forsythe SD, Pu T, Andrews SG, Madigan JP, Sadowski SM. Cancers (Basel). 2023 Jul 25; 15(15). Epub 2023 Jul 25.
- A wait-and-watch approach to small pancreatic neuroendocrine tumors: prognosis and survival.[Oncotarget. 2016]A wait-and-watch approach to small pancreatic neuroendocrine tumors: prognosis and survival.Massironi S, Rossi RE, Zilli A, Casazza G, Ciafardini C, Conte D. Oncotarget. 2016 Apr 5; 7(14):18978-83.
- Review Update on surgical treatment of pancreatic neuroendocrine neoplasms.[World J Gastroenterol. 2014]Review Update on surgical treatment of pancreatic neuroendocrine neoplasms.D'Haese JG, Tosolini C, Ceyhan GO, Kong B, Esposito I, Michalski CW, Kleeff J. World J Gastroenterol. 2014 Oct 14; 20(38):13893-8.
- Review Gastrointestinal Neuroendocrine Tumors and the Carcinoid Syndrome.[Endotext. 2000]Review Gastrointestinal Neuroendocrine Tumors and the Carcinoid Syndrome.Hofland J, de Herder WW. Endotext. 2000
- Whole-exome sequencing of calcitonin-producing pancreatic neuroendocrine neoplasms indicates a unique molecular signature.[Front Oncol. 2023]Whole-exome sequencing of calcitonin-producing pancreatic neuroendocrine neoplasms indicates a unique molecular signature.Döring C, Peer K, Bankov K, Bollmann C, Ramaswamy A, Di Fazio P, Wild PJ, Bartsch DK. Front Oncol. 2023; 13:1160921. Epub 2023 Sep 12.
- Pathophysiology and Treatment of Pancreatic Neuroendocrine Neoplasms (PNENS): Ne...Pathophysiology and Treatment of Pancreatic Neuroendocrine Neoplasms (PNENS): New Developments - Endotext
Your browsing activity is empty.
Activity recording is turned off.
See more...
