NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012-.

LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet].
Show detailsOVERVIEW
Introduction
Pyrazinamide is a first line antituberculosis medication, but is used only in combination with other antituberculosis medications such as isoniazid or rifampin. Pyrazinamide is associated with transient and asymptomatic elevations in serum aminotransferase levels and is a well known cause of clinically apparent, acute liver injury that can be severe and even fatal.
Background
Pyrazinamide (pir" a zin' a mide) is a synthetic pyrazine analogue of nicotinamide that has potent activity against mycobacterium tuberculosis. Its mechanism of action is unknown, but it appears to be both bactericidal and bacteriostatic. Pyrazinamide is considered an adjunctive therapy of tuberculosis and is always used in combination with another antimycobacterial agent such as isoniazid or rifampin. Pyrazinamide was developed and introduced into clinical use in 1956 and continues to be commonly used in therapy of active tuberculosis, but not for latent tuberculosis. It is available as tablets of 500 mg in several generic forms. A fixed combination of pyrazinamide (300 mg) with isoniazid (50 mg) and rifampin (120 mg) is also available under the brand name of Rifater. The recommended dosage of pyrazinamide is 15 to 30 mg/kg daily in one or more doses, but not in excess of 2 grams daily. Pyrazinamide can also be given in a twice weekly regimen of 50 to 70 mg/kg per dose allowing for observed administration and better compliance. Typically, pyrazinamide and ethambutol are given only for the first two months of 3- or 4-drug combination therapy of active tuberculosis, with continuation of isoniazid and rifampin for at least six months. Common side effects of pyrazinamide are fatigue, anorexia, gastrointestinal upset, arthralgias, rash and photosensitivity.
The management of tuberculosis is challenging and should be under the direction of physicians with expertise in tuberculosis therapy and management of its side effects. Optimal regimens of therapy for tuberculosis are complex and change frequently. Regularly updated recommendations on use of drugs for tuberculosis, including indications, contraindications, warnings, dosages and monitoring recommendations are available at the Centers for Disease Control and Prevention website: http://www.cdc.gov/tb/publications/guidelines/Treatment.htm.
Hepatotoxicity
Combination therapy for tuberculosis using pyrazinamide is commonly associated with transient and asymptomatic elevations in the serum aminotransferase levels. These elevations are usually less than five times the upper limit of the normal range. Because pyrazinamide is used only in combination with other antituberculosis medications, its contributions to serum enzyme elevations is not completely clear, but it is frequently incriminated in transient serum enzyme elevations. Clinically apparent liver disease with symptoms and jaundice also occur with pyrazinamide therapy and it is often considered the culprit in causing liver injury in the face of double or triple antituberculosis therapy (Case 1). Indeed, the use of a short, 2 month course of combination therapy with rifampin and pyrazinamide for latent tuberculosis was abandoned because of the frequency of severe liver injury with this regimen that was occasionally fatal (Case 2). The onset of injury due to pyrazinamide is generally after 4 to 8 weeks and occasionally becomes apparent only after the pyrazinamide is stopped. The pattern of liver enzyme elevations is typically hepatocellular and the clinical syndrome resembles acute viral hepatitis, much like isoniazid hepatotoxicity. Features of hypersensitivity (rash, fever and eosinophilia) are uncommon as are autoantibody formation. Liver biopsy demonstrates changes typical of acute hepatitis with portal and lobular inflammation, hepatocellular necrosis and variable degrees of cholestasis.
Likelihood score: A (well known cause of clinically apparent liver injury).
Mechanism of Injury
The mechanism of hepatic injury by pyrazinamide is not known, but the drug is extensively metabolized by the liver and injury may be caused by a metabolic intermediate. Hepatotoxicity from pyrazinamide appears to be more frequent with higher doses, suggesting a direct toxic effect, at least in part.
Outcome and Management
The appearance of liver injury with symptoms and serum aminotransferase levels above 3 times the ULN or any elevation in aminotransferase levels above 5 times the ULN should prompt withdrawal of pyrazinamide. Many cases of pyrazinamide hepatotoxicity are severe and prolonged, and fatal instances have occurred. Pyrazinamide has not been linked to chronic hepatitis or vanishing bile duct syndrome. There is no cross reactivity of the hepatic injury with other currently available antituberculosis agents.
After drug induced liver injury from combination antituberculosis therapy, reinitiation of therapy, if necessary, can be achieved best by sequential and incremental introduction of the agents (generally isoniazid and rifampicin), but that restarting several drugs at the standard dosage should be avoided particularly starting with pyrazinamide in full doses.
[First line medications used in the therapy of tuberculosis in the United States include isoniazid, pyrazinamide, ethambutol and the rifamycins including rifampin, rifabutin and rifapentine. Second line medications include streptomycin, capreomycin, cycloserine, ethionamide, bedaquiline, pretomanid, fluoroquinolones such as levofloxacin, and moxifloxacin, aminoglycosides such as amikacin, and para-aminosalicylic acid (PAS).]
Drug Class: Antituberculosis Agents
Other Drugs in the Class: Bedaquiline, Capreomycin, Cycloserine, Ethambutol, Ethionamide, Isoniazid, Pretomanid, Rifabutin, Rifampin, Rifapentine, Streptomycin
CASE REPORTS
Case 1. Acute liver failure due to combination antituberculosis therapy.(1)
A 60 year old woman with pulmonary tuberculosis was treated with isoniazid (300 mg daily), rifampin (600 mg daily) and pyrazinamide (1.5 gram daily) and improved rapidly, but was found to have jaundice with bilirubin 3.5 mg/dL, AST 538 U/L and alkaline phosphatase 148 U/L six weeks after starting therapy (Table). Her liver tests were reported to be normal before treatment. She had no history of liver disease, alcohol abuse, or risk factors for viral hepatitis. Isoniazid and rifampin were stopped but pyrazinamide continued. One week later, she was admitted to a local hospital for worsening hepatic function. She was mildly encephalopathic and the prothrombin time was prolonged. She was transferred to a liver transplant center. She was in stage 3 hepatic coma and required artificial ventilation. Pyrazinamide was stopped and ofloxacin, ethambutol and streptomycin started. Blood tests for hepatitis A, B and C were negative. She underwent successful liver transplantation 5 days after transfer and 3 weeks after onset of symptoms. She recovered uneventfully and was discharged 16 days after transplant on cyclosporine and prednisone. She was maintained on ethambutol, streptomycin and ofloxacin for tuberculosis, and sputum cultures remained negative.
Key Points
| Medication: | Isoniazid, rifampin, pyrazinamide |
|---|---|
| Pattern: | Hepatocellular (R=10.1) |
| Severity: | 5+ (acute liver failure requiring liver transplantation) |
| Latency: | 6 weeks |
| Recovery: | No |
| Other medications: | None mentioned |
Laboratory Values
| Time After Starting | Time After Stopping | AST* (U/L) | Protime* (Seconds) | Bilirubin* (mg/dL) | Other |
|---|---|---|---|---|---|
| Pre | Normal | Normal | Normal | ||
| Isoniazid, rifampin and pyrazinamide started for active tuberculosis | |||||
| 6 weeks | -7 days | 548 | 3.5 | ||
| 7 weeks | 0 | 1640 | 22.4 | 22.4 | Admission |
| 3 days | 1100 | 34.0 | 32.0 | Transfer | |
| 8 weeks | 7 days | 350 | 41.0 | 21.0 | |
| Emergency liver transplantation | |||||
| 3 months | 1 month | 30 | 11.0 | 5.0 | Discharge |
| Normal Values | <40 | <14 | <1.2 | ||
- *
Some values estimated from Figure 2.
Comment
This case demonstrates an acute viral hepatitis-like presentation of drug induced liver injury. While the case represents “definite” drug induced liver disease, it is impossible to be definitive about which agent was responsible. Isoniazid is a likely culprit, but pyrazinamide and (to a lesser degree) rifampin are also capable of causing acute severe liver injury, and continuing pyrazinamide in the face of an acute hepatitis-like syndrome was not advisable. Despite stopping isoniazid and rifampin therapy promptly, the patient progressed to hepatic failure and required liver transplantation within 3 weeks of initial presentation. Risk factors for antituberculosis therapy associated hepatitis included age and combination therapy. Risk factors for severe outcome were age and sex.
Case 2. Acute liver failure complicating two month course of rifampin and pyrazinamide.(2)
A 68 year old man with a history of a positive tuberculin test requested antituberculosis therapy so as to obviate the need for annual chest X-rays required by his employer. He had a history of alcoholic liver disease, but had stopped drinking 5 years previously. He also had gout, hypertension and type 2 diabetes for which he took aspirin, colchicine, ibuprofen, lisinopril and metformin on a chronic basis. He was started on rifampin (600 mg daily) and pyrazinamide (2 grams daily) and monitored monthly. He tolerated therapy without symptoms and liver tests were normal at baseline and at one month (Table). Immediately after stopping therapy, however, he began to feel ill and presented to the emergency room with jaundice, lethargy and confusion. On examination, he was icteric and had asterixis. Laboratory tests showed marked elevations in serum aminotransferase levels and bilirubin of 16.7 mg/dL. The prothrombin time was prolonged [INR 6.0]; and there were elevations in serum creatinine (5.4 mg/dL), ammonia (181 μmol/L: normal <35) and lactate (13.2 mmol/L, normal <2.2). Abdominal ultrasound showed ascites, but no evidence of biliary obstruction. He was given supportive care but developed worsening coma, respiratory and renal failure and died 3 days after admission.
Key Points
| Medication: | Pyrazinamide, rifampin |
|---|---|
| Pattern: | Hepatocellular (R=10) |
| Severity: | 5+ (death) |
| Latency: | 2 months |
| Recovery: | None |
| Other medications: | Aspirin, colchicine, ibuprofen, lisinopril, metformin |
Laboratory Values
| Time After Starting | Time After Stopping | ALT (U/L) | Alk P (U/L) | Bilirubin (mg/dL) | Other |
|---|---|---|---|---|---|
| Pre | 48 | 66 | 0.5 | INR=1.0 | |
| 0 | Pyrazinamide and rifampin started | ||||
| 2 weeks | 31 | 63 | 0.5 | ||
| 8 weeks | 0 | Pyrazinamide and rifampin stopped: asymptomatic | |||
| 3 days | 620 | 175 | 16.7 | INR=6.0 | |
| 9 weeks | 6 days | 1930 | 207 | 19.2 | INR=10.1 |
| 8 days | Death from multiorgan failure | ||||
| Normal Values | <45 | <130 | <1.2 | ||
Comment
The patient developed an abrupt onset of acute liver failure a few days after stopping an 8 week course of rifampin and pyrazinamide for latent tuberculosis. This combination is highly effective for latent tuberculosis, but has been associated with an unacceptably high rate of liver injury and cases of acute liver failure, for which reasons it is no longer recommended. Whether the injury was due to pyrazinamide or rifampin was unclear; there is likely an interaction that promotes the risk of injury. The risk of liver injury increases with age not just with isoniazid, but also with other antituberculosis medications and combinations. History of alcohol abuse and alcoholic liver injury are believed to also be risk factors for antituberculosis medication hepatotoxicity, but it has not been clearly defined. Because pyrazinamide inhibits the renal excretion of urates, it should be used with caution in patients with gout. This report does not mention testing for viral hepatitis or autopsy results; but the clinical history and course are entirely compatible with drug induced liver injury from antituberculosis medications.
PRODUCT INFORMATION
REPRESENTATIVE TRADE NAMES
Pyrazinamide – Generic, Tebrazid®
DRUG CLASS
Antituberculosis Agents
Product labeling at DailyMed, National Library of Medicine, NIH
CHEMICAL FORMULA AND STRUCTURE
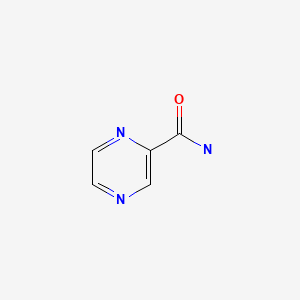
| DRUG | CAS REGISTRY NUMBER | MOLECULAR FORMULA | STRUCTURE |
|---|---|---|---|
| Pyrazinamide | 98-96-4 | C5-H5-N3-O |
|
CITED REFERENCES
- 1.
- Farrell FJ, Keeffe EB, Man KM, Imperial JC, Esquivel CO. Treatment of hepatic failure secondary to isoniazid hepatitis with liver transplantation. Dig Dis Sci. 1994;39:2255–9. [PubMed: 7924752]
- 2.
- Medinger A. Death associated with rifampin and pyrazinamide 2-month treatment of latent Mycobacterium tuberculosis. Chest. 2002;121:1710–2. [PubMed: 12006469]
ANNOTATED BIBLIOGRAPHY
References updated: 20 December 2020
Abbreviations: DRESS, drug rash with eosinophilia and systemic symptoms; HIV, human immunodeficiency virus; MAC, Mycobacterium avium complex; PAS, para-aminosalicylic acid.
- Zimmerman HJ. Antituberculosis agents. In, Zimmerman HJ. Hepatotoxicity: the adverse effects of drugs and other chemicals on the liver. 2nd ed. Philadelphia: Lippincott, 1999, pp. 611-21.(Extensive review of hepatotoxicity of antituberculosis medications including pyrazinamide published in 1999).
- Verma S, Kaplowitz N. Hepatotoxicity of antituberculosis drugs. In, Kaplowitz N, DeLeve LD, eds. Drug-induced liver disease. 3rd ed. Amsterdam: Elsevier, 2013, pp. 483-504.(Review of hepatotoxicity of antituberculosis drugs).
- Gumbo T. Chemotherapy of tuberculosis, mycobacterium avium complex disease and leprosy. In, Brunton LL, Hilal-Dandan R, Knollman BC, eds. Goodman & Gilman’s the pharmacological basis of therapeutics. 13th ed. New York: McGraw-Hill, 2018, pp. 1067-86.(Textbook of pharmacology and therapeutics).
- Aquinas SM. Reactions to antituberculosis drugs among Chinese in Hong Kong. Tubercle. 1964;45:181–7. [PubMed: 14227802](Among 389 patients treated with pyrazinamide and ethionamide for tuberculosis, side effects requiring discontinuation of therapy occurred in 4 [1%], including 3 with jaundice [all attributed to pyrazinamide], one of whom died).
- Ramakrishnan CV, Janardhanam B, Krishnamurthy DV, Stott H, Subbammal S, Tripathy SP. Toxicity of pyrazinamide, administered once weekly in high dosage, in tuberculous patients. Bull World Health Organ. 1968;39:775–9. [PMC free article: PMC2554433] [PubMed: 5306313](Pilot study of once weekly pyrazinamide [70 mg/kg] with isoniazid and streptomycin for 12 months in 19 patients with tuberculosis, found minor elevations in ALT and AST during therapy usually in first two weeks [peak ALT 63 U/L], but no patient developed clinically apparent hepatitis).
- Simpson AJ, Mirza AM, Martin JF, O'Brien TF Jr. Hepatitis secondary to pyrazinamide toxicity: accompaniments of transient hypolipoproteinemia, acanthocytosis, and changes in stomach and small bowel. South Med J. 1970;63:138–44. [PubMed: 5411920](34 year old developed jaundice 6 months after starting pyrazinamide and streptomycin for tuberculosis [bilirubin 26.4 mg/dL, AST 318 U/L, Alk P normal, protime 22.9 sec, cholesterol 68 mg/dL and acanthocytosis], resolving slowly over next 3 months).
- Rossouw JE, Saunders SJ. Hepatic complications of antituberculous therapy. Q J Med. 1975;44:1–16. [PubMed: 50605](Retrospective review identified 38 cases of hepatitis [~0.32%] due to antituberculosis therapy [Cape Town, SA], 16 due to PAS, 12 to PAS with isoniazid, 3 isoniazid alone, 1 each of others including ethambutol; rash and fever typical of PAS reactions, most within 90 days; 33 with jaundice).
- Hong Kong Tuberculosis Treatment services/British Medical Research Council. Adverse reactions to short-course regimens containing streptomycin, isoniazid, pyrazinamide and rifampicin in Hong Kong. Tubercle. 1976;57:81–95. [PubMed: 134476](Summary of 3 clinical trials of antituberculosis therapy [isoniazid, rifampin and pyrazinamide] from Hong Kong; hepatotoxicity in 6-11% of subjects, jaundice in 0.7%, no deaths, pyrazinamide implicated most frequently).
- Girling DJ. The hepatic toxicity of antituberculosis regimens containing isoniazid, rifampicin and pyrazinamide. Tubercle. 1978;59:13–32. [PubMed: 345572](History and review of hepatotoxicity of first line antituberculosis medications).
- Ellard GA, Mitchison DA, Girling DJ, Nunn AJ, Fox W. The hepatic toxicity of isoniazid among rapid and slow acetylators of the drug. Am Rev Respir Dis. 1978;118:628–9. [PubMed: 707886](Among 145 patients with active tuberculosis treated with isoniazid, pyrazinamide and streptomycin, there was no correlation of acetylator phenotype and ALT elevations; postulate that rapid acetylators also rapidly metabolize and excrete monoacetylhydrazine the first product of acetylation and purported hepatotoxin).
- Singapore Tuberculosis Service/British Medical Research Council. Clinical trial of six-month and four-month regimens of chemotherapy in the treatment of pulmonary tuberculosis. Am Rev Respir Dis. 1979;119:579–85. [PubMed: 375787](Among 397 patients with active tuberculosis treated with combination regimens, 11 [3%] developed hepatitis with jaundice, all in first 3 months, all recovered, all on rifampin, 10 on pyrazinamide and 2 on isoniazid).
- Addington WW. The side effects and interactions of antituberculosis drugs. Chest. 1979;76(6) Suppl:782–4. [PubMed: 510025](Review of side effects of antituberculosis medications states that isoniazid, rifampin and pyrazinamide are major causes of hepatotoxicity, but ethambutol rarely causes liver injury).
- Zierski M, Bek E. Side-effects of drug regimens used in short-course chemotherapy for pulmonary tuberculosis. A controlled clinical study. Tubercle. 1980;61:41–9. [PubMed: 6989067](Among 530 patients treated with 5 regimens for active tuberculosis and monitored monthly, 9% developed hepatic injury but usually without symptoms, ALT >250 U/L in 6 [1.1%], bilirubin rise in 7 [1.3%]).
- Pretet S, Perdrizet S. Rev Fr Mal Respir. 1980;8:307–30. [Toxicity of pyrazinamide in antituberculous treatments] French. [PubMed: 6454227](Review of large trials worldwide of antituberculosis therapy for rate of adverse drug reactions; overall hepatotoxicity occurred in 2.2% of 4,025 subjects treated with pyrazinamide containing regimens vs 0.5% of 2,368 without).
- British Thoracic Association. A controlled trial of six month chemotherapy in pulmonary tuberculosis. First report: results during chemotherapy. Br J Dis Chest. 1981;75:141–53. [PubMed: 7023526](Comparison of 3 regimens of therapy in 611 patients with active tuberculosis; 6 vs 9 months duration, with and without pyrazinamide; hepatitis occurred in 4% of patients with all 3 regimens and 8 patients [1.3%] had jaundice; hepatotoxicity occurred in 3% of men and 6% of women).
- Danan G, Pessayre D, Larrey D, Benhamou JP. Pyrazinamide fulminant hepatitis: an old hepatotoxin strikes again. Lancet. 1981;2:1056–7. [PubMed: 6118518](37 year old man with active tuberculosis developed elevated ALT after 8 months of therapy; isoniazid was stopped and pyrazinamide continued, and patient developed acute liver failure [ALT 30 times ULN, Alk P twice normal] and died).
- Pilheu JA, De Salvo MC, Koch O. Liver alterations in antituberculosis regimens containing pyrazinamide. Chest. 1981;80:720–2. [PubMed: 7307595](Prospective study of 59 alcoholic men with active tuberculosis treated with four drugs including isoniazid, rifampin, streptomycin and either pyrazinamide or ethambutol with monthly monitoring and liver biopsy before and after 2 months of therapy found no evidence of increased hepatotoxicity comparing pyrazinamide and ethambutol groups).
- Hong Kong Chest Service/British Medical Research Council. Controlled trial of 4 three-times-weekly regimens and a daily regimen all given for 6 months for pulmonary tuberculosis. Second report: the results up to 24 months. Tubercle. 1982;63:89–98. [PubMed: 6758252](Among 833 patients with active tuberculosis randomized to one of 5 regimens, hepatotoxicity occurred in 2.7% and only 3 [0.4%] were jaundiced; most were able to finish therapy).
- Girling DJ. Adverse effects of antituberculosis drugs. Drugs. 1982;23:56–74. [PubMed: 6459920](Review of side effects of drugs for tuberculosis; isoniazid alone leads to hepatitis in 0.5% of patients increasing with age from 0.3% <35, 0.8% >55 years, higher rates when it is combined with other agents; pyrazinamide found to be hepatotoxic in high doses [40 mg/kg/day], but not with lower doses – “So far there has been no report of a high incidence of hepatotoxicity with the modern pyrazinamide regimens studied”).
- Cohen CD, Sayed AR, Kirsch RE. Hepatic complications of antituberculosis therapy revisited. S Afr Med J. 1983;63:960–3. [PubMed: 6857425](Among 5565 patients treated for tuberculosis in Cape Town SA, 17 [0.3%] developed hepatitis, rate similar to that when PAS was used. Among 28 cases seen, 13 attributed to isoniazid, 16 pyrazinamide and 8 rifampin, mostly in combination; 2 deaths).
- Fiala W, Häcki MA, Brändli O. Schweiz Med Wochenschr. 1983;113:1956–9. [Pyrazinamide versus ethambutol in short-term therapy of lung tuberculosis. A randomized study] German. [PubMed: 6658432](Controlled trial comparing pyrazinamide [n=52: <2 g/day] vs ethambutol [n=61] for first 2-3 months with isoniazid and rifampin for 9 months to treat active tuberculosis; hepatitis occurred in 5 on pyrazinamide and 2 on ethambutol, all in first month, 3 symptomatic, no deaths, most were >70 years of age).
- Cataldi Amatriain RM, Rossi Case E. Rev Esp Enferm Apar Dig. 1984;66:174–7. [The hepatotoxicity of pyrazinamide] Spanish. [PubMed: 6494560](Pyrazinamide is similar in structure to isoniazid, initially found to be hepatotoxic, but then lower doses appeared to be safe and combination therapy allowed for shorter courses; among 34 patients treated, Alk P was 2-3 times ULN in 5 and ALT 2-3 times ULN in 4, all arising during first 30 days, all asymptomatic and resolving once pyrazinamide stopped).
- Cataldi Amatriain RM, Rossi Case E. Rev Esp Enferm Apar Dig. 1984;66:174–7. [The hepatotoxicity of pyrazinamide] Spanish. [PubMed: 6494560]
- Parthasarathy R, Sarma GR, Janardhanam B, Ramachandran P, Santha T, Sivasubramanian S, Somasundaram PR, Tripathy SP. Hepatic toxicity in South Indian patients during treatment of tuberculosis with short-course regimens containing isoniazid, rifampicin and pyrazinamide. Tubercle. 1986;67:99–108. [PubMed: 3775870](Hepatitis occurred in 10% of patients with spinal tuberculosis, 16-39% of children with meningitis and 2-8% with pulmonary disease treated with rifampin and isoniazid, arising 1-6 months after starting therapy, more commonly in slow acetylators).
- Tuberculosis Research Centre, Madras, and National Tuberculosis Institute, Bangalore. A controlled clinical trial of 3- and 5-month regimens in the treatment of sputum-positive pulmonary tuberculosis in South India. Am Rev Respir Dis. 1986;134:27–33. [PubMed: 3524334](Analysis of 3 regimens for therapy of active tuberculosis in 908 patients; jaundice occurred in 6-8% of subjects on rifampin, but only 1% of subjects on nonrifampin regimens [isoniazid, pyrazinamide and streptomycin], but no mention of rates of death or clinically apparent hepatitis).
- Swamy R, Acharyulu GS, Duraipandian M, Jawahar MS, Ramachandran R, Sarma GR. Liver function tests during treatment of tuberculosis with short-course regimens containing isoniazid, rifampicin & pyrazinamide. Indian J Med Res. 1987 Nov;86:549–57. [PubMed: 3451899]
- Donald PR, Schoeman JF, O'Kennedy A. Hepatic toxicity during chemotherapy for severe tuberculosis meningitis. Am J Dis Child. 1987;141:741–3. [PubMed: 2884866](Among 33 children with tuberculous meningitis treated with 3-4 agents, liver test abnormalities were common [85%], but usually mild and transient; only one child developed jaundice who was also IgM anti-HAV positive and who later tolerated therapy without recurrence).
- van Aalderen WMC, Knoester H, Knol K. Fulminant hepatitis during treatment with rifampicin, pyrazinamide and ethambutol. Eur J Pediatr. 1987;146:290–1. [PubMed: 3595648](10 year old girl developed nausea and ALT elevations 2 weeks after starting isoniazid, rifampin, pyrazinamide and ethambutol, resolving with stopping all drugs, but severe recurrence with fever, rash and fatal acute liver failure 6 weeks after adding pyrazinamide back to rifampin, ethambutol and kanamycin).
- Roden S, Lagneau M, Homasson JP. Rev Pneumol Clin. 1990;46:43. [Fulminant hepatitis induced by pyrazinamide] French. [PubMed: 2371480](68 year old woman developed jaundice 3.5 months after starting antituberculosis therapy with rifampin, pyrazinamide and ethambutol [bilirubin 9.6 mg/dL, ALT 1100 U/L, Alk P 185 U/L, prothrombin index 45%], with progressive hepatic failure leading to liver transplantation, by followed by death 10 days postoperatively).
- Kshirsagar NA, Karande SC, Potkar CN. A prospective survey of drug induced hepatotoxicity in a large hospital. Indian J Gastroenterol. 1992;11:13–5. [PubMed: 1551705](Among 11 cases of drug induced liver disease, 9 were due to antituberculosis medications, usually combinations of isoniazid, rifampin and pyrazinamide, 2 deaths).
- Padmini R, Srinivasan S, Nalini P, Mahadevan S. Short course chemotherapy for tuberculosis in children. J Trop Pediatr. 1993;39:361–4. [PubMed: 8133559](Among 83 children [<12 years] treated for tuberculosis [using 2-4 agents, including pyrazinamide] in India between 1988-91, 4 developed hepatitis with jaundice [bilirubin 2.0-4.5 mg/dL, ALT >4 times ULN], but all resolved with decrease in dose of isoniazid and rifampin).
- Altman C, Biour M, Grangé JD. Presse Med. 1993;22:1212–6. [Hepatic toxicity of antitubercular agents. Role of different drugs. 199 cases] French. [PubMed: 8248040](Analysis of 199 cases of hepatotoxicity from antituberculosis medications from literature [n=169] and French pharmacovigilance system [n=30]; overall mortality rate was 23%, rifampin cases had short latency [average 2 weeks] compared to isoniazid [11 weeks] and pyrazinamide [7 weeks] cases; there was no association with alcohol, but some with higher doses, particularly isoniazid).
- Robson SC, Pillans P, Willcox PA. Treatment of tuberculosis in patients with pre-existing liver disease or following hepatotoxic drug reactions. S Afr Med J. 1993;83:432–4. [PubMed: 8211464](Review of problems of treating tuberculosis in patients with preexisting liver disease, recommended avoiding pyrazinamide).
- van der Kooi K, Mottet JJ, Regamey C. Isoniazid is not always the cause of hepatitis during treatment of tuberculosis. Clin Infect Dis. 1994;19:987–8. [PubMed: 7893906](46 year old man developed hepatitis [ALT 900 U/L] 2 weeks after starting isoniazid, rifampin, pyrazinamide and ethambutol, with positive challenge with pyrazinamide and not by isoniazid; no mention of fever, rash or bilirubin levels).
- Türktaş H, Unsal M, Tülek N, Orüç O. Hepatotoxicity of antituberculosis therapy (rifampicin, isoniazid and pyrazinamide) or viral hepatitis. Tuber Lung Dis. 1994;75:58–60. [PubMed: 8161767](Among 705 Turkish adults with tuberculosis, 57 [8%] developed hepatitis with jaundice during therapy with isoniazid and rifampin; serologic testing showed hepatitis A in none, B in 6 and C in 4).
- Farrell FJ, Keeffe EB, Man KM, Imperial JC, Esquivel CO. Treatment of hepatic failure secondary to isoniazid hepatitis with liver transplantation. Dig Dis Sci. 1994;39:2255–9. [PubMed: 7924752](Two cases of acute liver failure attributed to isoniazid: 49 year old man on isoniazid for latent tuberculosis for 4 months developed jaundice [bilirubin 16.1 mg/dL, AST 2882 U/L]; and 60 year old woman developed jaundice 6 weeks after starting isoniazid, rifampin, and pyrazinamide for active tuberculosis [bilirubin 3.5 mg/dL, AST 548 U/L], both progressing to hepatic failure and undergoing successful liver transplantation).
- Horn DL, Hewlett D Jr, Alfalla C, Peterson S, Opal SM. Limited tolerance of ofloxacin and pyrazinamide prophylaxis against tuberculosis. N Engl J Med. 1994;330:1241. [PubMed: 8139647](Among 16 contacts of a case of multidrug resistant tuberculosis treated with pyrazinamide and ofloxacin, side effects were common; 4 had hepatitis [ALT 491-1776 U/L], only 2 completed therapy).
- Singh J, Arora A, Garg PK, Thakur VS, Pande JN, Tandon RK. Antituberculosis treatment-induced hepatotoxicity: role of predictive factors. Postgrad Med J. 1995;71:359–62. [PMC free article: PMC2398146] [PubMed: 7644398](Case control study of 60 patients with liver injury due to antituberculosis medications and 60 controls from India identified lower body mass index and use of pyrazinamide, but not age, sex, or acetylator status as risk factors).
- Mitchell I, Wendon J, Fitt S, Williams R. Anti-tuberculous therapy and acute liver failure. Lancet. 1995;345:555–6. [PubMed: 7786350](Four cases of acute liver failure in patients on isoniazid, rifampin and pyrazinamide; 3 women and 1 man, ages 31-61 years, jaundice after 1-6 weeks, two requiring liver transplantation, one recovered, one died; unclear which agent was responsible).
- Noble A. Antituberculous therapy and acute liver failure. Lancet. 1995 Apr 1;345:867. [PubMed: 7898260](Letter in response to Mitchell [1995] suggesting that patients be warned to stop the medication if they develop symptoms of liver injury).
- Janes SL, Behrens J. Antituberculous therapy and acute liver failure. Lancet. 1995;345:867. [PubMed: 7898259](Letter in response to Mitchell [1995] reporting a 45 year old man who developed hepatitis 5 weeks after starting isoniazid and rifampin [bilirubin 3.3 mg/dL, ALT 1884 U/L], who recovered rapidly with prompt stopping of both).
- Ozick LA, Jacob L, Comer GM, Lee TP, Ben-Zvi J, Donelson SS, Felton CP. Hepatotoxicity from isoniazid and rifampin in inner-city AIDS patients. Am J Gastroenterol. 1995;90:1978–80. [PubMed: 7485004](Among 70 AIDS patients with active tuberculosis receiving isoniazid, rifampin and pyrazinamide for 2 months, ALT or AST levels >200 U/L occurred in 8 [11%] with peak bilirubin values 0.6-2.9 mg/dL).
- Corbella X, Vadillo M, Cabellos C, Fernandez-Viladrich P, Rufi G. Hypersensitivity hepatitis due to pyrazinamide. Scand J Infect Dis. 1995;27:93–4. [PubMed: 7784827](59 year old woman developed abdominal pain and fever 1 month after starting isoniazid, rifampin, ethambutol and pyrazinamide [bilirubin 7.9 mg/dL, ALT 1260 U/L, 14% eosinophils], with positive rechallenge to pyrazinamide [ALT 3240 U/L], later tolerating ethambutol, rifampin and streptomycin).
- Moitinho E, Salmerón JM, Mas A, Bruguera M, Rodés J. Gastroenterol Hepatol. 1996;19:448–51. [Severe hepatotoxicity of tuberculostatic agents. Increase in the incidence] Spanish. [PubMed: 8998667](Among 27 cases of severe acute hepatitis seen in 1994 in Barcelona, 5 cases were due to antituberculosis medications, 1 woman and 4 men, ages 25 to 62 years, 2 with HBsAg, 1 with anti-HCV, arising after 11-62 days [peak bilirubin 8-41 mg/dL; ALT 450-2320 U/L, Alk P 1.5-2 times ULN], 3 died, one required liver transplant, one recovered spontaneously).
- de Souza AF. de Oliveira e Silva A, Baldi J, de Souza TN, Rizzo PM. Arq Gastroenterol. 1996;33:194–200. [Hepatic functional changes induced by the combined use of isoniazid, pyrazinamide and rifampicin in the treatment of pulmonary tuberculosis] Portuguese. [PubMed: 9302332]
- Pande JN, Singh SP, Khilnani GC, Khilnani S, Tandom RK. Risk factors for hepatotoxicity from antituberculosis drugs: a case-control study. Thorax. 1996;51:132–6. [PMC free article: PMC473016] [PubMed: 8711642](Comparison of 86 patients with hepatitis due to antituberculosis therapy to 406 patients who tolerated therapy; risk factors for liver injury were older age, history of high alcohol intake [20% vs 5%], more extensive disease [14% vs 3.5%], slow acetylator status [83% vs 64%] and use of pyrazinamide [63% vs 25%]).
- al Sarraf KA, Michielsen PP, Hauben EI, Lefebure A, Ramon AM, Van Marck EA, Pelckmans PA. Hepatotoxicity after a short course of low-dose pyrazinamide. Acta Gastroenterol Belg. 1996;59:251–3. [PubMed: 9085628](32 year old man with tuberculosis developed ALT elevations [50 times ULN] 10 weeks after starting isoniazid, resolving promptly on stopping, but rising again within 3 weeks of starting pyrazinamide and rifampin with worsening despite stopping both agents [peak bilirubin 1.2 mg/dL, ALT 1455 U/L, Alk P 107 U/L 10 weeks after withdrawal], resolving slowly and incompletely).
- Ormerod LP, Horsfield N. Frequency and type of reactions to antituberculosis drugs: observations in routine treatment. Tuber Lung Dis. 1996;77:37–42. [PubMed: 8733412](Among 1317 patients treated for active tuberculosis, hepatitis was attributed to rifampin in 1.4%, pyrazinamide in 1.2% and isoniazid in 0.3%, but none to ethambutol or streptomycin).
- Ormerod LP, Skinner C, Wales J. Hepatotoxicity of antituberculosis drugs. Thorax. 1996;51:111–3. [PMC free article: PMC473008] [PubMed: 8711637](Review of the problem of hepatotoxicity of antituberculosis medications and recommendations on monitoring, with biochemical monitoring recommended only for patients with preexisting liver disease; in the UK between 1965-86 there were 243 reports of liver injury due to antituberculosis therapy and 45 fatalities).
- Døssing M, Wilcke JT, Askgaard DS, Nybo B. Liver injury during antituberculosis treatment: an 11-year study. Tuber Lung Dis. 1996;77:335–40. [PubMed: 8796249](Retrospective chart review on 765 Danish patients treated for tuberculosis with 3 or 4 drugs for 6-9 months; 16% had AST elevations >2 times ULN usually during first month, but only 2% required modified regimen; 7 with jaundice, no fatalities; risk factors for hepatotoxicity were female sex, age and severe tuberculosis).
- Durand F, Jebrak G, Pessayre D, Fournier M, Bernuau J. Hepatotoxicity of antitubercular treatments. Rationale for monitoring liver status. Drug Saf. 1996;15:394–405. [PubMed: 8968694](Review and recommendations from France regarding monitoring of serum enzymes during therapy of tuberculosis; isoniazid may have direct hepatotoxicity because of dose relatedness and usual absence of recurrence on rechallenge; rifampin is rare cause of liver injury, usually with short latency period; pyrazinamide is clearly hepatotoxic at higher doses which should be kept to a minimum and given for 2 months only).
- Knobel B, Buyanowsky G, Dan M, Zaidel L. Pyrazinamide-induced granulomatous hepatitis. J Clin Gastroenterol. 1997;24:264–6. [PubMed: 9252856](52 year old developed fever 1 month after starting pyrazinamide and streptomycin for suspected tuberculosis [bilirubin 0.6 mg/dL, ALT 136 U/L, Alk P 752 U/L] without rash or eosinophilia, resolving on stopping, biopsy showing granulomas).
- Ridzon R, Meador J, Maxwell R, Higgins K, Weismuller P, Onorato IM. Asymptomatic hepatitis in persons who received alternative preventive therapy with pyrazinamide and ofloxacin. Clin Infect Dis. 1997;24:1264–5. [PubMed: 9195099](Among 22 contacts of patients with multidrug resistant tuberculosis given pyrazinamide and ofloxacin, 12 developed ALT elevations after 2-25 weeks resolving in 3-5 weeks of stopping; two above 35 years in age both developed elevations [ALT 1026 and 2,990 U/L], one had eosinophilia [35%] and jaundice [bilirubin 4.1 mg/dL]).
- Devoto FM, González C, Iannantuono R, Serra HA, González CD, Sáenz C. Risk factors for hepatotoxicity induced by antituberculosis drugs. Acta Physiol Pharmacol Ther Latinoam. 1997;47:197–202. [PubMed: 9504179]
- Hwang SJ, Wu JC, Lee CN, Yen FS, Lu CL, Lin TP, Lee SD. A prospective clinical study of isoniazid-rifampicin-pyrazinamide-induced liver injury in an area endemic for hepatitis B. J Gastroenterol Hepatol. 1997;12:87–91. [PubMed: 9076631](Prospective study of 240 patients treated for active tuberculosis with 3 drug regimen, found ALT elevations in 45% of 31 HBsAg carriers vs 26% of 209 controls; 1 death in a carrier, none in controls, but nevertheless concluded that HBsAg was not a risk factor for antituberculosis therapy associated liver injury).
- Ungo JR, Jones D, Ashkin D, Hollender ES, Bernstein D, Albanese AP, Pitchenik AE. Antituberculosis drug-induced hepatotoxicity. The role of hepatitis C virus and the human immunodeficiency virus. Am J Respir Crit Care Med. 1998;157:1871–6. [PubMed: 9620920](Among 134 patients with tuberculosis, 22 developed hepatotoxicity during therapy; risk factors were HCV infection [30% vs 11%] and HIV infection [27% vs 12%]; alpha interferon therapy of hepatitis C allowed for antituberculosis therapy without ALT elevations in some patients).
- Rose DN. Short-course prophylaxis against tuberculosis in HIV-infected persons. A decision and cost-effectiveness analysis. Ann Intern Med. 1998;129:779–86. [PubMed: 9841583](Cost-effectiveness analysis of various regimens for treating latent tuberculosis in HIV infected persons using Markov model and various predictions of efficacy, safety, tuberculosis development and survival used rates of fatal hepatitis of 0.002% found prophylaxis to be “greatly” beneficial, including regimens using pyrazinamide and rifampin for 2 months).
- Wada M, Yoshiyama T, Ogata H, Ito K, Mizutani S, Sugita H. Kekkaku. 1999;74:353–60. [Six-months chemotherapy (2HRZS or E/4HRE) of new cases of pulmonary tuberculosis—six year experiences on its effectiveness, toxicity, and acceptability] Japanese. [PubMed: 10355221]
- Corrigan D, Paton J. Hepatic enzyme abnormalities in children on triple therapy for tuberculosis. Pediatr Pulmonol. 1999;27:37–42. [PubMed: 10023790](Among 43 children prospectively monitored on rifampin, isoniazid and pyrazinamide therapy, 30% had abnormal liver tests, usually in first few weeks, 2 had symptoms, 1 jaundice; all patients continued or were restarted and finished therapy).
- Gordin F, Chaisson RE, Matts JP, Miller C, de Lourdes Garcia M, Hafner R, et al. Rifampin and pyrazinamide vs isoniazid for prevention of tuberculosis in HIV-infected persons: an international randomized trial. JAMA. 2000;283:1445–50. [PubMed: 10732934](Controlled trial in 1583 patients with HIV infection and latent tuberculosis comparing rifampin/pyrazinamide [R/P] for 2 months to isoniazid [INH] for 12 months; active tuberculosis arose in 0.8% per year of R/P vs 1.1% of INH treated; ALT elevations >10 times ULN occurred in 1.3% of R/P vs 3.3% of INH treated; one “grade 4 hepatitis” in R/P versus 2 in INH treated).
- Girotto L, Gjonovich A, Preciso G. Minerva Anestesiol. 2000;66:249–51. [Fulminant liver failure caused by antitubercular drugs. Report of a clinical case] Italian. [PubMed: 10832275](26 year old man with pulmonary tuberculosis treated with isoniazid, rifampin, ethambutol and pyrazinamide, presented after 1 month with fever, rash and jaundice [bilirubin 8.5 mg/dL, ALT 2757 U/L], renal insufficiency and hepatic and multiorgan failure; died after 10 days; autopsy showed massive liver necrosis and acute renal tubular necrosis).
- Meyers BR, Papanicolaou GA, Sheiner P, Emre S, Miller C. Tuberculosis in orthotopic liver transplant patients: increased toxicity of recommended agents; cure of disseminated infection with nonconventional regimens. Transplantation. 2000;69:64–9. [PubMed: 10653382](Toxicity was common in transplant patients treated for active tuberculosis using first line agents, but switching to second line agents was successful in treating the tuberculosis and well tolerated).
- Centers for Disease Control and Prevention (CDC). Fatal and severe hepatitis associated with rifampin and pyrazinamide for the treatment of latent tuberculosis infection—New York and Georgia, 2000. MMWR Morb Mortal Wkly Rep. 2001;50:289–91. [PubMed: 11330495](First alert: initial report of 2 cases of severe hepatitis during 2 month regimen of rifampin and pyrazinamide for latent tuberculosis; 53 year old man developed ALT elevation [1734 U/L] 33 days after starting regimen followed by jaundice [bilirubin 17.8 mg/dL] and fatal hepatic failure; 59 year old woman developed symptoms at end of therapy with bilirubin 11.4 mg/dL, ALT 1735 U/L, ANA 1:640, treated with prednisone and recovered; in both cases ALT monitoring did not prevent severe hepatitis).
- Centers for Disease Control and Prevention (CDC). Update: Fatal and severe liver injuries associated with rifampin and pyrazinamide for latent tuberculosis infection, and revisions in American Thoracic Society/CDC recommendations—United States, 2001. MMWR Morb Mortal Wkly Rep. 2001;50:733–5. [PubMed: 11787580](Second alert: during a 6 month period, 21 cases of liver injury were reported to the CDC associated with 2 month regimens of rifampin-pyrazinamide; 5 died of liver failure, ages 32 to 68 years, onset in 2nd month; recommended use of 9 months of isoniazid as standard regimen and need for close monitoring if rifampin-pyrazinamide is used).
- de Maria A, Berardi M, Dignetti P, Ferrera L, Viassolo L, Canonica GW. Side effects of antituberculosis treatment. Thorax. 2001;56:983. [PMC free article: PMC1745978] [PubMed: 11758512](Letter arguing against use of fixed dose combination pills of pyrazinamide with isoniazid and rifampin because of the risk of hepatotoxicity and inability to adjust relative doses).
- Burman WJ, Reves RR. Hepatotoxicity from rifampin plus pyrazinamide: lessons for policymakers and messages for care providers. Am J Respir Crit Care Med. 2001;164:1112–3. [PubMed: 11673194](Editorial in response to MMWR report on 21 cases of severe hepatotoxicity and 5 deaths due to the combination of rifampin and pyrazinamide).
- Vu D, Macdonald L. Antitubercular drugs (isoniazid, rifampin and pyrazinamide): hepatobiliary reactions. CMAJ. 2001;165:942–3, 946-7. [PubMed: 11599338](Review of 420 reports to the Canadian Monitoring Program of hepatotoxicity of antituberculosis drugs identified 258 due to isoniazid alone with 7 deaths; 27 to rifampin alone with 1 death; 110 to isoniazid and rifampin with 6 deaths; 25 related to pyrazinamide alone or in combination with 3 cases of death or hepatic failure; advises biochemical monitoring for patients above the age of 35).
- McCarthy M. US guidelines for treatment of latent tuberculosis revised. Lancet. 2001;358:816. [PubMed: 11564495](Newsletter on the modification of the US guidelines on treatment of latent tuberculosis, recommending avoidance of rifampin/pyrazinamide regimens).
- Tahaoğlu K, Ataç G, Sevim T, Tärün T, Yazicioğlu O, Horzum G, Gemci I, et al. The management of anti-tuberculosis drug-induced hepatotoxicity. Int J Tuberc Lung Dis. 2001;5:65–9. [PubMed: 11263519](Description of 45 patients with hepatotoxicity from antituberculosis therapy, ages 15 to 76 years, ALT 42-897 U/L, bilirubin 0.2-7.0 mg/dL, arising in 6-102 days, with resolution in 4-58 days. No recurrence in patients with gradual reintroduction of regimen without pyrazinamide compared to 6 cases [24%] in those with abrupt reintroduction).
- Wada M. Kekkaku. 2001;76:33–43. [Effectiveness and problems of PZA-containing 6-month regimen for the treatment of new pulmonary tuberculosis patients] Japanese. [PubMed: 11211781]
- Papastavros T, Dolovich LR, Holbrook A, Whitehead L, Loeb M. Adverse events associated with pyrazinamide and levofloxacin in the treatment of latent multidrug-resistant tuberculosis. CMAJ. 2002;167:131–6. [PMC free article: PMC117089] [PubMed: 12160118](Among 17 persons given pyrazinamide and levofloxacin for latent tuberculosis after contact with multidrug resistant tuberculosis, adverse reactions were common, 5 [29%] had ALT elevations [peak 80-504 U/L]; all recovered after stopping).
- Lee AM, Mennone JZ, Jones RC, Paul WS. Risk factors for hepatotoxicity associated with rifampin and pyrazinamide for the treatment of latent tuberculosis infection: experience from three public health tuberculosis clinics. Int J Tuberc Lung Dis. 2002;6:995–1000. [PubMed: 12475146](Among 148 patients with latent tuberculosis treated with rifampin/pyrazinamide, 14 [9.4%] had ALT elevations >5 times ULN, 12 were symptomatic [8%]; risk factors were female sex and recent infection).
- Medinger A. Death associated with rifampin and pyrazinamide 2-month treatment of latent Mycobacterium tuberculosis. Chest. 2002;121:1710–2. [PubMed: 12006469](68 year old man developed jaundice at end of 2 month course of rifampin and pyrazinamide for latent tuberculosis [bilirubin 19.2 mg/dL, ALT 1930 U/L, Alk P 207 U/L], with liver failure and death within 3 days of presentation: Case 2).
- Castro KG, Jereb JA, Koppaka VR, Cohn DL. Fatal liver injury associated with rifampin-pyrazinamide treatment of latent tuberculosis infection. Chest. 2003;123:967. [PubMed: 12628913](Letter in response to Medinger [2002] on need to report cases of hepatotoxicity of antituberculosis therapy to the CDC).
- Centers for Disease Control and Prevention (CDC). Update: Fatal and severe liver injuries associated with rifampin and pyrazinamide treatment for latent tuberculosis infection. MMWR Morb Mortal Wkly Rep. 2002;51:998–9. [PubMed: 12455909](Third alert: 40 cases of severe hepatotoxicity [8 fatal] associated with rifampin/pyrazinamide therapy of latent tuberculosis reported to CDC; issued revised guidelines cautioning against use of this regimen and only with no preexisting liver disease or alcohol use, with ALT monitoring and provision of drugs in two week increments).
- Jasmer RM, Saukkonen JJ, Blumberg HM, Daley CL, Bernardo J, Vittinghoff E, King MD, et al. Short-Course Rifampin and pyrazinamide for Tuberculosis Infection(SCRIPT) Study Investigators. Short-course rifampin and pyrazinamide compared with isoniazid for latent tuberculosis infection: a multicenter clinical trial. Ann Intern Med. 2002;137:640–7. [PubMed: 12379063](Controlled trial of isoniazid [INH: 6 months] vs rifampin and pyrazinamide [RP: 2 months] in 589 patients with latent tuberculosis, similar completion rates [57% vs 61%]; ALT rose >5 times ULN in 1% on INH vs 7.7% on RP; none resulted in hospitalization or death; no mention of jaundice).
- Ohkawa K, Hashiguchi M, Ohno K, Kiuchi C, Takahashi S, Kondo S, Echizen H, et al. Risk factors for antituberculous chemotherapy-induced hepatotoxicity in Japanese pediatric patients. Clin Pharmacol Ther. 2002;72:220–6. [PubMed: 12189369](Retrospective analysis of 99 children who received therapy for tuberculosis, 8 developed hepatotoxicity; risk factors identified were young age and pyrazinamide exposure).
- Teleman MD, Chee CB, Earnest A, Wang YT. Hepatotoxicity of tuberculosis chemotherapy under general programme conditions in Singapore. Int J Tuberc Lung Dis. 2002;6:699–705. [PubMed: 12150482](Retrospective analysis of 1036 patients treated for active tuberculosis in Singapore during 1998 found 55 cases of liver injury [5.3%], 37 symptomatic [3.6%], 18 jaundiced [1.8%], 3 died [0.3%: all on pyrazinamide]; 48 able to restart therapy; risk factors were age >60 years and baseline liver test abnormalities).
- Centers for Disease Control and Prevention (CDC). American Thoracic Society. Update: adverse event data and revised American Thoracic Society/CDC recommendations against the use of rifampin and pyrazinamide for treatment of latent tuberculosis infection—United States, 2003. MMWR Morb Mortal Wkly Rep. 2003;52:735–9. [PubMed: 12904741](Fourth alert and issuing of recommendations against use of rifampin/pyrazinamide for latent tuberculosis; with this regimen, estimated rate of ALT elevations >5 times ULN was 2.6%, hospitalization for hepatitis 0.3% and death 0.09%).
- American Thoracic Society. Centers for Disease Control and Prevention (CDC). Infectious Diseases Society of America. Treatment of tuberculosis. MMWR Recomm Rep. 2003;52(RR-11):1–77. [PubMed: 12836625](Detailed recommendations on therapy of tuberculosis including drug regimens, side effects, monitoring and optimal approaches to follow up).
- Kunimoto D, Warman A, Beckon A, Doering D, Melenka L. Severe hepatotoxicity associated with rifampin-pyrazinamide preventative therapy requiring transplantation in an individual at low risk for hepatotoxicity. Clin Infect Dis. 2003;36:e158–61. [PubMed: 12802781](37 year old man had AST elevations at end of 2 month course of rifampin/pyrazinamide, with levels rising thereafter to AST 2409 U/L and bilirubin 7.8 mg/dL and need for liver transplant 2 months later).
- Nagayama N, Masuda K, Baba M, Tamura A, Nagai H, Akagawa S, Kawabe Y, et al. Kekkaku. 2003;78:339–46. [Secular increase in the incidence rate of drug-induced hepatitis due to anti-tuberculosis chemotherapy including isoniazid and rifampicin] Japanese. [PubMed: 12739393](In Japan there was an apparent increase in rate of ALT elevations reported during isoniazid and rifampin therapy between the years 1980-83 [9%] and 1998-2000 [27%]).
- Yee D, Valiquette C, Pelletier M, Parisien I, Rocher I, Menzies D. Incidence of serious side effects from first-line antituberculosis drugs among patients treated for active tuberculosis. Am J Respir Crit Care Med. 2003;167:1472–7. [PubMed: 12569078](Among 408 adult patients treated for tuberculosis, 37 [9%] had 46 serious adverse events including 12 [3%] instances of hepatitis [ALT >5 times ULN]; risk factors were age [hazard ratio 4.8-7.7], female sex [2.2] and Asian birthplace [2.2]; hepatitis arose in 2% on pyrazinamide and 1% on isoniazid).
- Mohan A, Sharma SK. Side effects of antituberculosis drugs. Am J Respir Crit Care Med. 2004;169:882–3. [PubMed: 15044223](Letter in response to Yee [2003] requesting further information on doses and clinical features; reply by authors stating that high rate of hepatotoxicity in comparison to reports from India was not due to higher doses of pyrazinamide, but may have been partially due to patient age and diverse ethnic background of patients).
- McNeill L, Allen M, Estrada C, Cook P. Pyrazinamide and rifampin vs isoniazid for the treatment of latent tuberculosis: improved completion rates but more hepatotoxicity. Chest. 2003;123:102–6. [PubMed: 12527609](Between 1999-2002, 110 patients were treated with pyrazinamide/rifampin [P/R] for 2 months and 114 with isoniazid [INH] for 6 months for latent tuberculosis; completion rates were higher [71% vs 59%] as was hepatotoxicity [13% vs 4%] with PR than INH, and 2 patients had clinically apparent hepatitis [ALT 45-67 times ULN at 4 weeks] but both survived; after intensive monitoring, no further severe cases on P/R).
- Stout JE, Engemann JJ, Cheng AC, Fortenberry ER, Hamilton CD. Safety of 2 months of rifampin and pyrazinamide for treatment of latent tuberculosis. Am J Respir Crit Care Med. 2003;167:824–7. [PubMed: 12446275](Among 114 patients receiving 2 month course of rifampin/pyrazinamide, 67% completed therapy and 6 had hepatitis [5.3%] but all resolved; no hospitalizations or death).
- Fernández-Villar A, Sopeña B, Fernández-Villar J, Vázquez-Gallardo R, Ulloa F, Leiro V, Mosteiro M, et al. The influence of risk factors on the severity of anti-tuberculosis drug-induced hepatotoxicity. Int J Tuberc Lung Dis. 2004;8:1499–505. [PubMed: 15636498](Among 471 patients receiving antituberculosis therapy, 56 [12%] developed ALT elevations >3 times ULN, 16 [3.4%] and symptoms and 5 [1%] were jaundiced; no deaths. Rates of hepatotoxicity higher in patients with risk factors than without [18.2% vs 5.6%]).
- Jasmer RM, Snyder DC, Saukkonen JJ, Hopewell PC, Bernardo J, King MD, Kawamura LM, et al. Short-course rifampin and pyrazinamide compared with isoniazid for latent tuberculosis infection: a cost-effective analysis based on a multicenter clinical trial. Clin Infect Dis. 2004;38:363–9. [PubMed: 14727206](Cost effectiveness analysis of two regimens of therapy for latent tuberculosis, suggesting that isoniazid alone for 9 months is less expensive than a short course of rifampin and pyrazinamide and has similar long term efficacy [both regimens increased life expectancy by 1.2 years]).
- Ijaz K, McElroy PD, Navin TR. Short-course rifampin and pyrazinamide compared with isoniazid for latent tuberculosis infection: a cost-effectiveness analysis based on a multicenter clinical trial. Clin Infect Dis. 2004;39:289. [PubMed: 15307044](Letter in response to Jasmer [2004], stressing the recommendation that rifampin and pyrazinamide not be used to treat latent tuberculosis, a 9 month course of isoniazid being safer and more cost effective).
- Gordin FM, Cohn DL, Matts JP, Chaisson RE, O'Brien RJ., Terry Beirn Community Programs for Clinical Research on AIDS. Adult AIDS Clinical Trials Group; Centers for Disease Control and Prevention. Hepatotoxicity of rifampin and pyrazinamide in the treatment of latent tuberculosis infection in HIV-infected persons: is it different than in HIV-uninfected persons? Clin Infect Dis. 2004;39:561–5. [PubMed: 15356822](Detailed reanalysis of results of randomized controlled trial comparing 12 months of isoniazid [INH] to 2 months of rifampin/pyrazinamide [R/P] for latent tuberculosis in HIV infected patients; comparing INH to RP recipients, bilirubin >2.5 mg/dL occurred in 0.6% vs 1.8% and AST >250 U/L in 1.6% vs 2.1%, but no hospitalizations or deaths due to hepatotoxicity occurred in the trial; older age was only risk factor identified).
- van Hest R, Baars H, Kik S, van Gerven P, Trompenaars MC, Kalisvaart N, Keizer S, et al. Hepatotoxicity of rifampin-pyrazinamide and isoniazid preventive therapy and tuberculosis treatment. Clin Infect Dis. 2004;39:488–96. [PubMed: 15356811](Retrospective analysis of patients starting preventive antituberculosis therapy; ALT elevations above 5 times ULN occurred in 8.4% of those receiving 2 months of rifampin/pyrazinamide vs 3.4% of those receiving 6 months of isoniazid).
- Marra F, Cox VC, FitzGerald JM, Moadebi S, Elwood RK. Successful treatment of multidrug-resistant tuberculosis following drug-induced hepatic necrosis requiring liver transplant. Int J Tuberc Lung Dis. 2004;8:905–9. [PubMed: 15260286](Patient with tuberculous lymphadenitis was treated with isoniazid, rifampin, ethambutol and pyrazinamide and was switched to ciprofloxacin with pyrazinamide and ethambutol when resistance testing was done; four days later she developed fever, rash and fatigue [bilirubin normal, ALT 285 U/L, Alk P normal], but then worsened [bilirubin 15.2 mg/dL, ALT 1165 U/L, Alk P 141 U/L], ultimately requiring liver transplant; she was later treated successfully with levofloxacin, amikacin and streptomycin).
- Campos-Franco J, Gonzalez-Quintela A, Alende-Sixto MR. Isoniazid-induced hyperacute liver failure in a young patient receiving carbamazepine. Eur J Intern Med. 2004;15:396–7. [PubMed: 15522577](16 year old girl on long term carbamazepine and clobazam with ileocecal tuberculosis developed hepatitis and drowsiness with tremor, 5 days after starting isoniazid, rifampin and pyrazinamide [bilirubin 3.2 mg/dL, ALT 658 U/L, prothrombin index 12%], resolving spontaneously despite continuing anticonvulsants and later tolerating reintroduction of rifampin and pyrazinamide).
- Sharma SK. Antituberculosis drugs and hepatotoxicity. Infect Genet Evol. 2004;4:167–70. [PubMed: 15157635](Review of hepatotoxicity of isoniazid, rifampin and pyrazinamide with focus on role of acetylator status).
- Kandula NR, Dworkin MS, Carroll MR, Lauderdale DS. Tuberculosis prevention in Mexican immigrants: limitations of short-course therapy. Am J Prev Med. 2004;26:163–6. [PubMed: 14751331](Among 34 immigrants and contacts of a patient with multidrug resistant tuberculosis treated with rifampin/pyrazinamide, 1 of 11 children [9%] and 4 of 23 adults [17%] developed hepatotoxicity [ALT 165-547 U/L] after 10-53 days; all recovered and were able to finish 4 months of rifampin; points out impracticality of biochemical monitoring in immigrant population).
- McElroy PD, Ijaz K, Lambert LA, Jereb JA, Iademarco MF, Castro KG, Navin TR. National survey to measure rates of liver injury, hospitalization, and death associated with rifampin and pyrazinamide for latent tuberculosis infection. Clin Infect Dis. 2005;41:1125–33. [PubMed: 16163632](Survey of 110 health care programs using the 2 month rifampin/pyrazinamide regimen for latent tuberculosis in 8087 patients between 2000-2; ALT elevations >5 times ULN occurred in 2.4% and hepatitis in 1.9% with 23 hospitalizations and 7 deaths [0.1%] due to acute liver injury; higher than historical rates with isoniazid).
- Cook PP. Rifampin and pyrazinamide for treatment of latent tuberculosis infection. Clin Infect Dis. 2006;42:892–author reply 892-3. [PubMed: 16477576](Letter in response to McElroy [2005] questioning use of ALT for AST values used to define hepatotoxicity rates; reply by authors suggesting use of “AT” to indicate both enzymes).
- Younossian AB, Rochat T, Ketterer JP, Wacker J, Janssens JP. High hepatotoxicity of pyrazinamide and ethambutol for treatment of latent tuberculosis. Eur Respir J. 2005;26:462–4. [PubMed: 16135729](Among 12 persons treated with pyrazinamide and ethambutol for latent tuberculosis and contact with a patient with multidrug resistant disease, 7 developed liver injury [ALT 82-1338 U/L], after 87-247 days, 3 with symptoms, no mention of bilirubin levels).
- Potolidis E, Mantadakis E, Zeniodi MH, Samonis G. Rifampin plus pyrazinamide-induced hepatitis requiring hospitalization in a 30-y-old male with latent tuberculosis. Scand J Infect Dis. 2005;37:155–7. [PubMed: 15773037](30 year old man developed nausea and dehydration 6 weeks after starting rifampin and pyrazinamide for latent tuberculosis [bilirubin normal, ALT “in the range of 600 to 2000” U/L], resolving within 3 weeks of stopping).
- Lee BH, Koh WJ, Choi MS, Suh GY, Chung MP, Kim H, Kwon OJ. Inactive hepatitis B surface antigen carrier state and hepatotoxicity during antituberculosis chemotherapy. Chest. 2005;127:1304–11. [PubMed: 15821209](Retrospective case control study of 110 HBsAg carriers and 97 controls from Korea who received 3-4 drug antituberculosis therapy; any ALT elevations occurred in 34% of carriers vs 20% of controls and were >3 times ULN in 8% vs 4%; no risk factors identified, most tolerated reintroduction of therapy without pyrazinamide).
- Lobato MN, Reves RR, Jasmer RM, Grabau JC, Bock NN, Shang N. 2RZ Study Group. Adverse events and treatment completion for latent tuberculosis in jail inmates and homeless persons. Chest. 2005;127:1296–303. [PubMed: 15821208](Analysis of 2 month course of rifampin and pyrazinamide in jail and homeless populations found ALT elevations >5 times ULN in 6% of patients, one of whom died of acute liver failure, risk factors were older age and baseline ALT levels).
- Idilman R, Ersoz S, Coban S, Kumbasar O, Bozkaya H. Antituberculous therapy-induced fulminant hepatic failure: successful treatment with liver transplantation and nonstandard antituberculous therapy. Liver Transpl. 2006;12:1427–30. [PubMed: 16933231](19 year old woman with peritoneal tuberculosis developed jaundice 4 days after starting isoniazid, rifampin, ethambutol and pyrazinamide [bilirubin 10.5 mg/dL, ALT 1332 U/L, protime 71 sec], undergoing living donor liver transplantation within 2 days and afterwards treated with streptomycin, ethambutol and cycloserine with no recurrence).
- Yew WW, Leung CC. Antituberculosis drugs and hepatotoxicity. Respirology. 2006;11:699–707. [PubMed: 17052297](Review of incidence, causes, risk factors and management of hepatotoxicity of antituberculosis medications).
- Senaratne WV, Pinidiyapathirage MJ, Perera GA, Wickremasinghe AR. Anti-tuberculosis drug induced hepatitis – a Sri Lankan experience. Ceylon Med J. 2006;51:9–14. [PubMed: 16898030](Among 783 patients treated for active tuberculosis, 9.5% developed hepatitis; major risk factor being older age).
- Cook PP, Maldonado RA, Yarnell CT, Holbert D. Safety and completion rate of short-course therapy for treatment of latent tuberculosis infection. Clin Infect Dis. 2006;43:271–5. [PubMed: 16804838](Retrospective analysis of 459 patients treated in public health departments for latent tuberculosis; completion rates were 78% for 2 months of rifampin/pyrazinamide vs 66% for 9 months of isoniazid; any ALT elevations in 18% vs 11%; ALT >5 times ULN in 6% vs 2%; no deaths).
- Schechter M, Zajdenverg R, Falco G, Barnes GL, Faulhaber JC, Coberly JS, Moore RD, et al. Weekly rifapentine/isoniazid or daily rifampin/pyrazinamide for latent tuberculosis in household contacts. Am J Respir Crit Care Med. 2006;173:922–6. [PMC free article: PMC2662911] [PubMed: 16474028](In trial comparing rifapentine with isoniazid for 3 months [n=206] vs rifampin and pyrazinamide for 2 months [n=193]; hepatotoxicity arose in 10% on pyrazinamide vs 1% on isoniazid combination, all resolved within two months, no hospitalizations or deaths).
- Forget EJ, Menzies D. Adverse reactions to first-line antituberculosis drugs. Expert Opin Drug Saf. 2006;5:231–49. [PubMed: 16503745](Review of side effects including hepatotoxicity of isoniazid, rifampin, ethambutol and pyrazinamide; overall rate of hepatotoxicity from pyrazinamide is 1.3-2.5%).
- Ijaz K, Jereb JA, Lambert LA, Bower WA, Spradling PR, McElroy PD, Iademarco MF, et al. Severe or fatal liver injury in 50 patients in the United States taking rifampin and pyrazinamide for latent tuberculosis infection. Clin Infect Dis. 2006;42:346–55. [PubMed: 16392079](Analysis of 50 cases of severe hepatotoxicity from rifampin/pyrazinamide therapy of latent tuberculosis occurring in US between 1998-2004 and arising during or within 1 month of stopping therapy; fatality rate higher in older patients and with later onset; patients frequently on other potentially hepatotoxic medications).
- Shigeto E., Committee for Treatment Japanese Society for Tuberculosis. Kekkaku. 2007;82:467–73. [Survey of anti-tuberculosis drug-induced severe liver injury in Japan] Japanese. [PubMed: 17564126](Abstract: Survey questionnaire to 114 Japanese hospitals identified 70 cases of severe liver injury and 8 deaths due to antituberculosis therapy between 1994-2003).
- Sabaté M, Ibáñez L, Pérez E, Vidal X, Buti M, Xiol X, Mas A, et al. Risk of acute liver injury associated with the use of drugs: a multicentre population survey. Aliment Pharmacol Ther. 2007;25:1401–9. [PubMed: 17539979](Population based survey of 126 cases of acute liver injury due to drugs between 1993-1999 in Spain calculated relative risk of injury compared to the general population to be 1300 with use of triple therapy [isoniazid, rifampin and pyrazinamide] and 154 for isoniazid alone, ranking first and third).
- Marra F, Marra CA, Bruchet N, Richardson K, Moadebi S, Elwood RK, Fitzgerald JM. Adverse drug reactions associated with first-line anti-tuberculosis drug regimens. Int J Tuberc Lung Dis. 2007;11:868–75. [PubMed: 17705952](Among 1061 patients treated for active tuberculosis in British Columbia between 2000-2005 [usually with 3-4 drugs for ~8 months], 148 [14%: 2% per month] developed hepatitis defined by ALT >5 times ULN or symptoms and ALT >3 times ULN]; independent risk factors were pyrazinamide, female sex, older age and baseline ALT levels, but not HBV, HCV or race).
- Kwon YS, Koh WJ, Suh GY, Chung MP, Kim H, Kwon OJ. Hepatitis C virus infection and hepatotoxicity during antituberculosis chemotherapy. Chest. 2007;131:803–8. [PubMed: 17356096](Retrospective analysis of 54 patients with HCV infection and 97 controls receiving therapy for active tuberculosis; ALT >3 times ULN occurred in 13% of HCV infected vs 4% of controls; none died or required hospitalization).
- Huang YS. Genetic polymorphisms of drug-metabolizing enzymes and the susceptibility to antituberculosis drug-induced liver injury. Expert Opin Drug Metab Toxicol. 2007;3:1–8. [PubMed: 17269890](Two gene variants have been linked to an increased risk of hepatotoxicity of antituberculosis medications; NAT2 and CYP 2E1, but the associations require further confirmation).
- Aouam K, Chaabane A, Loussaïef C, Ben Romdhane F, Boughattas NA, Chakroun M. Med Mal Infect. 2007;37:253–61. [Adverse effects of antitubercular drugs: epidemiology, mechanisms, and patient management] French. [PubMed: 17336011](Review of toxicities of antituberculosis agents; pyrazinamide is the most likely agent to cause side effects which can include hepatocellular injury, which is more common with higher doses and arising within the first 2 months of therapy).
- Marzuki OA, Fauzi AR, Ayoub S, Kamarul Imran M. Prevalence and risk factors of anti-tuberculosis drug-induced hepatitis in Malaysia. Singapore Med J. 2008;49:688–93. [PubMed: 18830542](Among 473 patients treated for tuberculosis, 46 [9.7%] developed ALT elevations >3 times ULN; in a case control analysis, concurrent HIV infection was a risk factor).
- Makhlouf HA, Helmy A, Fawzy E, El-Attar M, Rashed HA. A prospective study of antituberculous drug-induced hepatotoxicity in an area endemic for liver diseases. Hepatol Int. 2008;2:353–60. [PMC free article: PMC2716885] [PubMed: 19669265](Among 100 Middle Eastern patients treated for active tuberculosis, 15% had liver toxicity arising in 15-60 days, resolving in 15-45 days of stopping; bilirubin >2 in 5%, 1 death; able to restart therapy in all).
- Leiro V, Fernández-Villar A, Valverde D, Constenla L, Vázquez R, Piñeiro L, González-Quintela A. Influence of glutathione S-transferase M1 and T1 homozygous null mutations on the risk of antituberculosis drug-induced hepatotoxicity in a Caucasian population. Liver International. 2008;28:835–9. [PubMed: 18397238](Case control study of 35 cases of hepatotoxicity from antituberculosis therapy and 60 controls; found higher rate of T1 GST variants in cases [49%] than controls [27%]; no difference for M1 variants [34% vs 42%]).
- Possuelo LG, Castelan JA, de Brito TC, Ribeiro AW, Cafrune PI, Picon PD, Santos AR, et al. Association of slow N-acetyltransferase 2 profile and anti-TB drug-induced hepatotoxicity in patients from Southern Brazil. Eur J Clin Pharmacol. 2008;64:673–81. [PubMed: 18421452](Among 17 persons given pyrazinamide and levofloxacin for latent tuberculosis after contact with multidrug resistant tuberculosis, adverse reactions were common, 5 [29%] had ALT elevations [peak 80-504 U/L]; all recovered).
- Chang KC, Leung CC, Yew WW, Lau TY, Tam CM. Hepatotoxicity of pyrazinamide: cohort and case-control analyses. Am J Respir Crit Care Med. 2008;177:1391–6. [PubMed: 18388355](Among 3007 patients starting antituberculosis therapy over a six month period, 150 [5.0%] developed ALT elevations >3 times ULN including 48 that arose after 3 months; in a case control analysis, risk factors were hepatitis B and C and regimens with pyrazinamide).
- Vieira DE, Gomes M. Adverse effects of tuberculosis treatment: experience at an outpatient clinic of a teaching hospital in the city of São Paulo, Brazil. J Bras Pneumol. 2008;34:1049–55. [PubMed: 19180340](Among 297 patients treated for tuberculosis between 2000 and 2006, 24 [10.6%] had hepatotoxicity, 7 [2.4%] symptomatic, no deaths).
- Yimer G, Aderaye G, Amogne W, Makonnen E, Aklillu E, Lindquist L, Yamuah L, et al. Anti-tuberculosis therapy-induced hepatotoxicity among Ethiopian HIV-positive and negative patients. PLoS One. 2008;3:e1809. [PMC free article: PMC2265547] [PubMed: 18350147](Between 2004-5, 103 HIV-positive and 94 HIV-negative patients were treated for tuberculosis; subclinical hepatotoxicity occurred in 17% and clinical apparent liver injury with jaundice in 4.1%; risk factors were HIV-positivity and other drug intake, but not age, sex, hepatitis or body mass index).
- Sun F, Chen Y, Xiang Y, Zhan S. Drug-metabolising enzyme polymorphisms and predisposition to anti-tuberculosis drug-induced liver injury: a meta-analysis. Int J Tuberc Lung Dis. 2008;12:994–1002. [PubMed: 18713495](Prospective study in 261 patients treated for tuberculosis; 16% developed hepatitis [ALT or AST > times 5 times ULN or >3 times with symptoms/jaundice], predictive factors were preexisting elevations or low albumin; 49% had at least one ALT or AST elevation, 25% >2 times, 17% >3 times and 10% >5 times ULN).
- Kaneko Y, Nagayama N, Kawabe Y, Shimada M, Suzuki J, Kunogi M, Matsui Y, et al. Kekkaku. 2008;83:13–9. [Drug-induced hepatotoxicity caused by anti-tuberculosis drugs in tuberculosis patients complicated with chronic hepatitis] Japanese. [PubMed: 18283910](Abstract: there was a substantial increase in frequency of ALT elevations in patients with chronic hepatitis C associated with pyrazinamide therapy).
- P V K. Palaian S, Ojha P, P R S. Pattern of adverse drug reactions experienced by tuberculosis patients in a tertiary care teaching hospital in Western Nepal. Pak J Pharm Sci. 2008;21:51–6. [PubMed: 18166520](Abstract: Retrospective analysis of 326 Nepalese patients treated for tuberculosis identified 24 [7.4%] with AST elevations; no fatalities from liver disease).
- Tostmann A, Boeree MJ, Aarnoutse RE, de Lange WC, van der Ven AJ, Dekhuijzen R. Antituberculosis drug-induced hepatotoxicity: concise up-to-date review. J Gastroenterol Hepatol. 2008;23:192–202. [PubMed: 17995946](Review of incidence, pathogenesis, clinical course, risk factors and management of drug induced liver disease due to antituberculosis medications).
- Centers for Disease Control and Prevention(CDC). Trends in tuberculosis—United States, 2008. MMWR Morb Mortal Wkly Rep. 2009;58:249–53. [PubMed: 19300406](In 2008, 12,898 cases of active tuberculosis in US were reported, lowest rate since reporting began in 1953; incidence rate=3/100,000; 1.2% with multidrug resistant strains).
- Chalasani N, Fontana RJ, Bonkovsky HL, Watkins PB, Davern T, Serrano J, Yang H, Rochon J., Drug Induced Liver Injury Network (DILIN). Causes, clinical features, and outcomes from a prospective study of drug-induced liver injury in the United States. Gastroenterology. 2008;135:1924–34. [PMC free article: PMC3654244] [PubMed: 18955056](Among 300 cases of drug induced liver disease in the US collected between 2004 and 2008; isoniazid accounted for 13 cases [4%, ranking 2nd in frequency], pyrazinamide 1, rifampin 1, and isoniazid combined with other agents 2).
- Semfke A, Wackernagel C, Vier H, Schütz A, Wiechmann V, Gillissen A. Histologically proven isoniazid hepatoxicity in complicated tuberculous salpingitis. Ther Adv Respir Dis. 2009;3:159–62. [PubMed: 19723821](49 year old woman with suspected tuberculous salpingitis developed fever and ALT elevations 1 month after starting isoniazid, rifampin and pyrazinamide, resolved only when she stopped isoniazid, later tolerating rifampin).
- Ho CC, Chen YC, Hu FC, Yu CJ, Yang PC, Luh KT. Safety of fluoroquinolone use in patients with hepatotoxicity induced by anti-tuberculosis regimens. Clin Infect Dis. 2009;48:1526–33. [PubMed: 19400686](Among 1191 patients treated for active tuberculosis, 134 [11%] developed liver injury [risk factors were baseline ALT levels and pyrazinamide use]; then treated with ethambutol with or without streptomycin and randomized to also receive levofloxacin or moxifloxacin, only 1 in each group continued to have ALT elevations and most were then able to restart isoniazid and rifampin).
- Walker NF, Kliner M, Turner D, Bhagani S, Cropley I, Hopkins S, Lipman M. Hepatotoxicity and antituberculosis therapy: time to revise UK guidance? Thorax. 2009;64:918. [PubMed: 19786720](During a one year period, 14 of 94 patients [15%] with active tuberculosis developed ALT elevations >3 times ULN during therapy; more common in HIV-positive [35%] than -negative [7%]; recommended routine HIV screening and biochemical monitoring in high risk groups).
- Tostmann A, van den Boogaard J, Semvua H, Kisonga R, Kibiki GS, Aarnoutse RE, Boeree MJ. Antituberculosis drug-induced hepatotoxicity is uncommon in Tanzanian hospitalized pulmonary TB patients. Trop Med Int Health. 2010;15:268–72. [PubMed: 20409289](Among 112 Tanzanian patients with tuberculosis monitored during first 2 months of therapy with isoniazid, rifampin, pyrazinamide and ethambutol, only 7 [6.3%] had ALT elevations [peak level 87 U/L], but none required dose modification).
- Sharma SK, Singla R, Sarda P, Mohan A, Makharia G, Jayaswal A, Sreenivas V, et al. Safety of 3 different reintroduction regimens of antituberculosis drugs after development of antituberculosis treatment-induced hepatotoxicity. Clin Infect Dis. 2010;50:833–9. [PubMed: 20156055](175 patients with hepatic injury [ALT or AST >5 times ULN or symptoms with ALT elevations] attributed to antituberculosis medications were randomized to 3 different approaches to restarting therapy after initial episode resolved; 11% had recurrence with rates similar for all three regimens [starting agents at full doses or gradual escalation], onset in 5-35 days, none fatal and few with jaundice).
- Saukkonen J. Challenges in reintroducing tuberculosis medications after hepatotoxicity. Clin Infect Dis. 2010;50:840–2. [PubMed: 20156056](Editorial in response to Sharma [2010], discussing the paradox of why the injury does not recur more often, whether rechallenge should be with one agent at a time, and whether such rechallenge is warranted in patients with severe hepatotoxicity).
- Pasipanodya JG, Gumbo T. Clinical and toxicodynamic evidence that high-dose pyrazinamide is not more hepatotoxic than the low doses currently used. Antimicrob Agents Chemother. 2010;54:2847–54. [PMC free article: PMC2897291] [PubMed: 20439617](The investigators conducted a systematic review of the literature and metaanalysis; findings indicated lack of association between high dose/exposure and liver toxicity; they concluded that attributable toxicity is idiosyncratic).
- Ferrajolo C, Capuano A, Verhamme KM, Schuemie M, Rossi F, Stricker BH, Sturkenboom MC. Drug-induced hepatic injury in children: a case/non-case study of suspected adverse drug reactions in VigiBase. Br J Clin Pharmacol. 2010;70:721–8. [PMC free article: PMC2997312] [PubMed: 21039766](Worldwide pharmacovigilance database contained 9036 hepatic adverse drug reactions in children, 2 antituberculosis agents were among the top 40 cases, including isoniazid [24th, 47 cases], and rifampin [35th, 37 cases]).
- Reuben A, Koch DG, Lee WM., Acute Liver Failure Study Group. Drug-induced acute liver failure: results of a U.S. multicenter, prospective study. Hepatology. 2010;52:2065–76. [PMC free article: PMC3992250] [PubMed: 20949552](Among 1198 patients with acute liver failure enrolled in a US prospective study between 1998 and 2007, 133 were attributed to drug induced liver injury and 25 to antituberculosis agents, including 15 to isoniazid alone [ranking first], 6 to isoniazid combined with other agents, 3 to rifampin and pyrazinamide and 1 to dapsone).
- Devarbhavi H, Dierkhising R, Kremers WK, Sandeep MS, Karanth D, Adarsh CK. Single-center experience with drug-induced liver injury from India: causes, outcome, prognosis, and predictors of mortality. Am J Gastroenterol. 2010;105:2396–404. [PubMed: 20648003](Among 313 cases of drug induced liver injury seen between 1997 and 2008 at a large hospital in Bangalore, India, 181 [58%] were attributed to antituberculosis agents which accounted for 39 of 54 [72%] fatal cases).
- Molleston JP, Fontana RJ, Lopez MJ, Kleiner DE, Gu J, Chalasani N. Drug-induced Liver Injury Network. Characteristics of idiosyncratic drug-induced liver injury in children: results from the DILIN prospective study. J Pediatr Gastroenterol Nutr. 2011;53:182–9. [PMC free article: PMC3634369] [PubMed: 21788760](Among 30 children with suspected drug induced liver injury, 3 were attributed to isoniazid).
- Coca NS, Oliveira MS, Voieta I, Antunes CM, Lambertucci JR. Antituberculosis drug-induced hepatotoxicity: a comparison between patients with and without human immunodeficiency virus seropositivity. Rev Soc Bras Med Trop. 2010;43:624–8. [PubMed: 21181011](Using different definitions, rates of hepatotoxicity during antituberculosis therapy were either the same or higher among HIV infected than noninfected subjects).
- Ichai P, Saliba F, Antoun F, Azoulay D, Sebagh M, Antonini TM, Escaut L, Delvart V, Castaing D, Samuel D. Acute liver failure due to antitubercular therapy: Strategy for antitubercular treatment before and after liver transplantation. Liver Transpl. 2010;16:1136–46. [PubMed: 20879012](Description of 14 patients with acute liver failure due to antituberculosis therapy presenting between 1986 and 2008, including 4 men and 10 women, ages 17 to 64 years, on therapy for 1 week to 9 months with isoniazid alone [n=1] or in combination with rifampin, pyrazinamide and ethambutol [bilirubin 1.8 to 39 mg/dL, ALT 214-2020], 7 recovering spontaneously, 6 undergoing liver transplantation, and one dying without transplant).
- Baniasadi S, Eftekhari P, Tabarsi P, Fahimi F, Raoufy MR, Masjedi MR, Velayati AA. Protective effect of N-acetylcysteine on antituberculosis drug-induced hepatotoxicity. Eur J Gastroenterol Hepatol. 2010;22:1235–8. [PubMed: 20461008](Among 60 patients started on antituberculosis therapy using 4 drugs with or without concurrent N-acetylcysteine [NAC: 600 mg twice daily], hepatotoxicity arose in 37% of controls vs 0% of NAC treated subjects, the injury usually arising within the first week and resolving within 8 days of stopping).
- Lobue P, Menzies D. Treatment of latent tuberculosis infection: An update. Respirology. 2010;15:603–22. [PubMed: 20409026](Extensive review of the efficacy and safety of various regimens used in the treatment of latent tuberculosis).
- Leung CC, Rieder HL, Lange C, Yew WW. Treatment of latent infection with Mycobacterium tuberculosis: update 2010. Eur Respir J. 2011;37:690–711. [PubMed: 20693257](Review of the efficacy, adherence rates, cost effectiveness and safety of various regimens for the therapy of latent tuberculosis).
- Nader LA, de Mattos AA, Picon PD, Bassanesi SL, De Mattos AZ, Pineiro Rodriguez M. Hepatotoxicity due to rifampicin, isoniazid and pyrazinamide in patients with tuberculosis: is anti-HCV a risk factor? Ann Hepatol. 2010;9:70–4. [PubMed: 20308724](Retrospective analysis of 534 patients treated for tuberculosis between 1998 and 2006 found that HIV positivity and high doses of isoniazid were independently associated with a higher risk of hepatotoxicity).
- Donald PR. Antituberculosis drug-induced hepatotoxicity in children. Pediatr Rep. 2011;3:e16. [PMC free article: PMC3133498] [PubMed: 21772953](Extensive review of the literature on hepatotoxicity of antituberculosis medications in children concluded that liver injury occurs in children with isoniazid, pyrazinamide and rifampin, but at a lower rate than in adults).
- Thongraung W, Sittidach M, Khwansuwan P, Sariyasuntorn K, Wongsampan S. Evaluation of the physicians' approach to the diagnosis and treatment of patients with antituberculosis drug-induced hepatotoxicity. J Eval Clin Pract. 2012;18:1119–25. [PubMed: 21696520](Survey of physician practices in Southern Thailand found variability in diagnosis and management of tuberculosis which were not always in compliance with published guidelines).
- Perriot J, Chambonnet E, Eschalier A. Rev Mal Respir. 2011;28:542–55. [Managing the adverse events of antitubercular agents] French. [PubMed: 21549908](Review of the side effects of antituberculosis medications).
- Devarbhavi H. Antituberculous drug-induced liver injury: current perspective. Trop Gastroenterol. 2011;32:167–74. [PubMed: 22332331](Review of hepatotoxicity of antituberculosis medications).
- Leiro-Fernandez V, Valverde D, Vázquez-Gallardo R, Botana-Rial M, Constenla L, Agúndez JA, Fernández-Villar A. N-acetyltransferase 2 polymorphisms and risk of anti-tuberculosis drug-induced hepatotoxicity in Caucasians. Int J Tuberc Lung Dis. 2011;15:1403–8. [PubMed: 22283902](Analysis of N-acetyltransferase 2 polymorphisms in 50 Spanish patients with ALT elevations on antituberculosis therapy and in 67 controls found no difference in acetylation status).
- Donald PR. Antituberculosis drug-induced hepatotoxicity in children. Pediatr Rep. 2011;3:e16. [PMC free article: PMC3133498] [PubMed: 21772953](Review of the hepatotoxicity and antituberculosis medications in children; rates of hepatotoxicity are lower in children than adults, ALT elevations occurring in 10% and jaundice in 0.8% of those receiving antituberculosis therapy and at lower rates with chemo-prophylaxis).
- Whitney J, Hurwitz M, Mojtahed A, Hwang C, Gallo A. Acute liver failure in a pediatric patient with disseminated tuberculosis. Dig Dis Sci. 2011;56:2780–3. [PubMed: 21695402](7 year old boy developed jaundice and mental confusion 2 months after starting therapy with isoniazid, pyrazinamide, rifampin, ethambutol, fluconazole and ceftriaxone [bilirubin 6.5 mg/dL, ALT 396 U/L, GGT 207 U/L, INR 3.6], resolving spontaneously after stopping the regimen and switching to amikacin, cycloserine, ethambutol, levofloxacin and linezolid).
- Kim SH, Kim SH, Yoon HJ, Shin DH, Park SS, Kim YS, Park JS, et al. TNF-α genetic polymorphism -308G/A and antituberculosis drug-induced hepatitis. Liver Int. 2012;32:809–14. [PubMed: 22151084](Analysis of polymorphisms of TNF alpha in 77 Korean patients who developed ALT elevations >3 times ULN on antituberculosis therapy and 229 who did not , found slightly higher rate of variant allele [-308G/A] associated with hepatotoxicity [26% vs 15%]).
- Malla I, Fauda M, Casanueva E, Fernández MI, Amante M, Cheang Y, Giacove G, et al. Arch Argent Pediatr. 2012;110:e35–8. [Fulminant hepatic failure due to tuberculostatic drugs: case report] Spanish. [PubMed: 22760756](10 year old girl developed jaundice 40 days after starting isoniazid, pyrazinamide and rifampin [bilirubin 13 mg/dL, ALT 37 times ULN, INR 8.5], developing progressive hepatic failure and undergoing successful liver transplantation 2 days after admission).
- Cai Y, Yi J, Zhou C, Shen X. Pharmacogenetic study of drug-metabolising enzyme polymorphisms on the risk of anti-tuberculosis drug-induced liver injury: a meta-analysis. PLoS One. 2012;7:e47769. [PMC free article: PMC3474796] [PubMed: 23082213](Metaanalysis of studies of genetic polymorphisms and hepatotoxicity of drugs for tuberculosis identified 38 publications including 2225 patients and 4906 controls; modest association was found between slow NAT2 acetylation genotypes and glutathione-S-transferase genotypes and hepatotoxicity, but largely in Asian populations).
- Zhou Y, Yang L, Liao Z, He X, Zhou Y, Guo H. Epidemiology of drug-induced liver injury in China: a systematic analysis of the Chinese literature including 21,789 patients. Eur J Gastroenterol Hepatol. 2013;25:825–9. [PubMed: 23510965](Systematic review of the Chinese literature on drug induced liver injury from 1994 to 2011 identified 1119 reports on 21,789 patients; antituberculosis agents were the most commonly implicated drugs [31%]).
- Nomi F, Hosaka K, Kurosawa T. Kekkaku. 2013 Sep;88(9):647–51. [A clinical investigation of seven patients with pulmonary tuberculosis who developed mixed liver injury during oral anti-tuberculosis treatment] Japanese. [PubMed: 24298690](Among 321 patients with tuberculosis treated at a single referral hospital during a 5 year period, 7 [2%] developed liver injury with a mixed pattern of enzyme elevations, onset averaging 28 days and resulting in fatality in one).
- Kato H, Horita N, Miyazawa N, Yoshiyama T, Ueda A, Ishigatsubo Y. Risk factors for liver injury with an elevated serum bilirubin concentration caused by antituberculous drugs. Intern Med. 2013;52:2209–14. [PubMed: 24088753](In a retrospective analysis of potential risk factors for liver injury with jaundice due to antituberculosis agents, the level of activity of daily living and concurrent cardiac disease was found to be most strongly associated with occurrence of liver injury).
- Miyazawa N, Horita N, Tomaru K, Tsukahara T, Takahashi R, Sasaki M, Ishigatsubo Y. Kekkaku. 2013;88:297–300. [Comparison of drug-induced hepatitis occurring in elderly and younger patients during anti-tuberculosis treatment with a regimen including pyrazinamide] [PubMed: 23672170](Among 36 Japanese patients with tuberculosis treated with isoniazid, pyrazinamide, rifampin and ethambutol, liver injury occurred in 12.5% over 80 and 14% less than 80 years of age).
- Björnsson ES, Bergmann OM, Björnsson HK, Kvaran RB, Olafsson S. Incidence, presentation and outcomes in patients with drug-induced liver injury in the general population of Iceland. Gastroenterology. 2013;144:1419–25. [PubMed: 23419359](In a population based study of drug induced liver injury from Iceland, 96 cases were identified over a 2 year period, 2 [2%] of which were attributed to anti-tuberculosis agents, 1 isoniazid and 1 rifampin).
- Gupta VH, Singh M, Amarapurkar DN, Sasi P, Joshi JM, Baijal R. Association of GST null genotypes with anti-tuberculosis drug induced hepatotoxicity in Western Indian population. Ann Hepatol. 2013;12:959–65. H R PK, et al. [PubMed: 24114827](Among 296 patients being treated for tuberculosis, 17% developed ALT elevations during therapy and evidence of liver injury was associated with homozygous null mutations at both glutathione S transferase M1 and T1 gene loci).
- Li C, Long J, Hu X, Zhou Y. GSTM1 and GSTT1 genetic polymorphisms and risk of anti-tuberculosis drug-induced hepatotoxicity: an updated meta-analysis. Eur J Clin Microbiol Infect Dis. 2013;32:859–68. [PubMed: 23377313](Metaanalysis of studies on the association of tuberculosis drug associated liver injury and polymorphisms of the glutathione S-transferase genes found weak associations only).
- Kumar N, Kedarisetty CK, Kumar S, Khillan V, Sarin SK. Antitubercular therapy in patients with cirrhosis: challenges and options. World J Gastroenterol. 2014;20:5760–72. [PMC free article: PMC4024786] [PubMed: 24914337](Review of the frequency, pathogenesis and management of hepatotoxicity of antituberculosis therapy in patients with preexisting cirrhosis, who appear to have an increased risk of liver injury from these agents as well as an increased risk of severe outcomes).
- Shin HJ, Lee HS, Kim YI, Lim SC, Jung JP, Ko YC, Kwon YS. Hepatotoxicity of anti-tuberculosis chemotherapy in patients with liver cirrhosis. Int J Tuberc Lung Dis. 2014;18:347–51. [PubMed: 24670574](Among 50 patients with cirrhosis and 147 controls without cirrhosis who had active tuberculosis and were treated with isoniazid, rifampin ethambutol and pyrazinamide, abnormal liver tests arose in 72% of cirrhotics vs 46% of controls, and predefined "hepatotoxicity" arose in 8% vs 3%, but no patient required hospitalization or died).
- Huang YS. Recent progress in genetic variation and risk of antituberculosis drug-induced liver injury. J Chin Med Assoc. 2014;77:169–73. [PubMed: 24593909](Critical review of studies on genetic variations associated with antituberculosis drug induced liver injury focusing on N-acetyltransferase (NAT), CYP 2E1, glutathione S transferase and manganese superoxide dismutase, the strongest association being with NAT and slow acetylator status).
- Satyaraddi A, Velpandian T, Sharma SK, Vishnubhatla S, Sharma A, Sirohiwal A, Makharia GK, et al. Correlation of plasma anti-tuberculosis drug levels with subsequent development of hepatotoxicity. Int J Tuberc Lung Dis. 2014;18:188–95. i-iii. [PubMed: 24429311](Among 110 patients being treated for tuberculosis, 15 developed evidence of liver injury [enzyme or bilirubin elevations or symptoms]; plasma free rifampin, but not isoniazid or pyrazinamide, levels were higher in patients who developed liver injury than in those who did not).
- Xiang Y, Ma L, Wu W, Liu W, Li Y, Zhu X, Wang Q, et al. The incidence of liver injury in Uyghur patients treated for TB in Xinjiang Uyghur autonomous region, China, and its association with hepatic enzyme polymorphisms nat2, cyp2e1, gstm1 and gstt1. PLoS One. 2014;9:e85905. [PMC free article: PMC3900431] [PubMed: 24465778](Among 2244 patients treated for tuberculosis, 89 [4%] had aminotransferase elevations above 2 times and 0.4% above 5 times ULN after 2 months of treatment; evidence of liver injury was more common in persons with NAT2*5*CT genotype, slow acetylator phenotype, but not with CYP 2E1 polymorphisms).
- Hernández N, Bessone F, Sánchez A, di Pace M, Brahm J, Zapata R, A, Chirino R, et al. Profile of idiosyncratic drug induced liver injury in Latin America. An analysis of published reports. Ann Hepatol. 2014;13:231–9. [PubMed: 24552865](Systematic review of literature of drug induced liver injury in Latin American countries published from 1996 to 2012 identified 176 cases, 13 [7%] due to antituberculosis agents including 6 to isoniazid, 1 to pyrazinamide, 1 to rifampin and 5 to combinations).
- Li X, Liu Y, Zhang E, He Q, Tang YB. Liver transplantation in antituberculosis drugs-induced fulminant hepatic failure: a case report and review of the literature. Medicine (Baltimore). 2015;94:e1665. [PMC free article: PMC5008466] [PubMed: 26656321](39 year old Chinese woman developed jaundice 3 months after starting isoniazid, rifampin and pyrazinamide for latent tuberculosis [bilirubin 0.6 rising to 41.1 mg/dL, ALT 1414 U/L, Alk P xxx U/L, protime 71 sec], with progressive hepatic failure undergoing liver transplantation 4 months after onset complicated by acute and ultimately fatal chronic rejection).
- Jeong I, Park JS, Cho YJ, Yoon HI, Song J, Lee CT, Lee JH. Drug-induced hepatotoxicity of anti-tuberculosis drugs and their serum levels. J Korean Med Sci. 2015;30:167–72. [PMC free article: PMC4310943] [PubMed: 25653488](Among 195 Korean patients with tuberculosis treated with a 4-drug regimen of isoniazid, rifampin, ethambutol and pyrazinamide, drug levels taken within the first week of treatment did not correlate with subsequent rate or severity of antituberculosis drug induced liver injury [n=12: 6%]).
- Sharma P, Tyagi P, Singla V, Bansal N, Kumar A, Arora A. Clinical and biochemical profile of tuberculosis in patients with liver cirrhosis. J Clin Exp Hepatol. 2015;5:8–13. [PMC free article: PMC4415191] [PubMed: 25941429](Among 67 patients with cirrhosis treated for tuberculosis with isoniazid, rifampin, ethambutol and either orofloxacin or pyrazinamide, evidence of drug induced liver injury arose in 35% of cases, but none were attributed to ethambutol or pyrazinamide and there were no deaths attributed to drug toxicity).
- Horita N, Miyazawa N, Yoshiyama T, Kojima R, Ishigatsubo Y, Kaneko T. Currently used low-dose pyrazinamide does not increase liver-injury in the first two months of tuberculosis treatment. Intern Med. 2015;54:2315–20. [PubMed: 26370854](Among 383 Chinese patients with tuberculosis treated with isoniazid, rifampin and ethambutol with or without pyrazinamide analyzed in a retrospective study, liver injury arising within the first two months of therapy was less frequent with four drug [8%] than 3-drug regimen [24%]).
- Chalasani N, Bonkovsky HL, Fontana R, Lee W, Stolz A, Talwalkar J, Reddy KR, et al. United States Drug Induced Liver Injury Network. Features and outcomes of 899 patients with drug-induced liver injury: The DILIN Prospective Study. Gastroenterology. 2015;148:1340–52.e7. [PMC free article: PMC4446235] [PubMed: 25754159](Among 899 cases of drug induced liver injury enrolled in a US prospective study between 2004 and 2013, 54 [6%] were attributed to antituberculosis agents including 48 to isoniazid, 2 to rifampin, 2 [0.2%] to pyrazinamide, 1 to ethambutol and 1 to isoniazid and rifampin).
- Bright-Thomas RJ, Gondker AR, Morris J, Ormerod LP. Drug-related hepatitis in patients treated with standard anti-tuberculosis chemotherapy over a 30-year period. Int J Tuberc Lung Dis. 2016;20:1621–4. [PubMed: 27931337](Among 2070 patients with tuberculosis treated over a 30 year period at a single UK referral center, 63 [3%] developed hepatitis requiring discontinuation of therapy of whom two died; the hepatitis rate was higher in whites than Asians and in females than males, and occurred more commonly with older age; 57% of cases were attributed to pyrazinamide, 32% to rifampin and 11% to isoniazid).
- Ji GY, Wang Y, Wu SQ, Liu QQ, Wu JC, Zhang MM, Sandford AJ, et al. Association between TXNRD1 polymorphisms and anti-tuberculosis drug-induced hepatotoxicity in a prospective study. Genet Mol Res. 2016;15:27706680. [PubMed: 27706680](Among 248 patients with treated for tuberculosis, 24 [10%] developed evidence of drug induced liver injury, among whom thioredoxin reductase 1 gene variants [involved resistance to oxidative damage] were no different in distribution than among those who did not develop liver injury).
- Guaoua S, Ratbi I, El Bouazzi O, Hammi S, Tebaa A, Bourkadi JE, Bencheikh RS, et al. NAT2 genotypes in Moroccan patients with hepatotoxicity due to antituberculosis drugs. Genet Test Mol Biomarkers. 2016;20:680–4. [PubMed: 27541622](Among 42 Moroccan patients with liver injury during antituberculosis therapy, there were no differences in frequency of acetylator status or in NAT2 genotypes compared to controls).
- Wang FJ, Wang Y, Niu T, Lu WX, Sandford AJ, He JQ. Update meta-analysis of the CYP2E1 RsaI/PstI and DraI polymorphisms and risk of antituberculosis drug-induced hepatotoxicity: evidence from 26 studies. J Clin Pharm Ther. 2016;41:334–40. [PubMed: 27062377](Metaanalysis of 26 studies involving 7423 patients found that the CYP2E1 RsaI/PstI c1/c1 polymorphism was associated with an increased risk for liver injury caused by antituberculosis therapy with an odds ratio of 1.32).
- Lin HS, Cheng CW, Lin MS, Chou YL, Chang PJ, Lin JC, Ye JJ. The clinical outcomes of oldest old patients with tuberculosis treated by regimens containing rifampicin, isoniazid, and pyrazinamide. Clin Interv Aging. 2016;11:299–306. [PMC free article: PMC4795580] [PubMed: 27042029](Among 700 Taiwanese patients with active tuberculosis treated with isoniazid, rifampin and pyrazinamide, hepatitis arose in 83 [12%] and was more frequent in those above the age of 80 years [17.4%] compared to younger [10.2%], but details on severity and clinical features were not provided).
- Chen G, Wu SQ, Feng M, Wang Y, Wu JC, Ji GY, Zhang MM, et al. Association of UGT2B7 polymorphisms with risk of induced liver injury by anti-tuberculosis drugs in Chinese Han. Int J Immunopathol Pharmacol. 2017;30:434–8. [PMC free article: PMC5806809] [PubMed: 28934901](Among 247 Chinese patients who received antituberculosis therapy for at least 3 months, 24 [10%] developed evidence of hepatic injury, but there was no association with polymorphisms of UGT2B7 a major hepatic enzyme responsible for glucuronidation and excretion of drugs and xenobiotics).
- Zenner D, Beer N, Harris RJ, Lipman MC, Stagg HR, van der Werf MJ. Treatment of latent tuberculosis infection: an updated network meta-analysis. Ann Intern Med. 2017;167:248–55. [PubMed: 28761946](A metaanalysis of 61 studies on regimens for therapy of "latent tuberculosis" found evidence of lower rates of hepatotoxicity with rifampin only or short courses of isoniazid and rifampin compared to longer isoniazid only regimens).
- Wang Y, Xiang X, Wu SQ, Chen G, Zhang MM, Wang MG, Wang FJ, et al. Association of CYP2B6 gene polymorphisms and anti-tuberculosis drug-induced hepatotoxicity in a Chinese population. Infect Genet Evol. 2017;51:198–202. [PubMed: 28389387](Among 343 Chinese patients treated for tuberculosis, several CYP2B6 gene polymorphisms were found to be associated with a reduced risk for hepatotoxicity).
- Angamo MT, Chalmers L, Curtain CM, Yilma D, Bereznicki L. Mortality from adverse drug reaction-related hospitalizations in south-west Ethiopia: A cross-sectional study. J Clin Pharm Ther. 2018;43:790–8. [PubMed: 29722039](Among 1001 patients admitted to an Ethiopian hospital over a 1 year period, 103 were admitted for an adverse drug reaction, 15 of which were fatal, with 7 due to liver failure, implicated agents being isoniazid, pyrazinamide, efavirenz and tenofovir).
- Lee LN, Huang CT, Hsu CL, Chang HC, Jan IS, Liu JL, Sheu JC, et al. Mitochondrial DNA variants in patients with liver injury due to anti-tuberculosis drugs. J Clin Med. 2019;8:1207. [PMC free article: PMC6723168] [PubMed: 31412578](Among 38 patients who developed liver injury during antituberculosis therapy, sequencing of mitochondrial DNA identified a higher frequency of point mutations in complex I NADH subunits 1 and 5 in isoniazid cases [n=8] but not in pyrazinamide [n=14] or rifampin [n=16] associated cases).
- Hagiwara E, Suido Y, Asaoka M, Katano T, Okuda R, Sekine A, Kitamura H, et al. Safety of pyrazinamide-including regimen in late elderly patients with pulmonary tuberculosis: A prospective randomized open-label study. J Infect Chemother. 2019;25:1026–30. [PubMed: 31229376](Among 89 elderly patients [80 years or older] with tuberculosis treated with isoniazid, rifampin and ethambutol with or without pyrazinamide, rates of discontinuation were similar between the two groups [33% vs 35%] as well as rates of ALT or AST elevations above 2.5 times ULN [18% vs 23%]).
- Wang Y, Xiang X, Huang WW, Sandford AJ, Wu SQ, Zhang MM, Wang MG, et al. Association of PXR and CAR polymorphisms and antituberculosis drug-induced hepatotoxicity. Sci Rep. 2019;9:2217. [PMC free article: PMC6379441] [PubMed: 30778091](Among 502 patients with tuberculosis treated with isoniazid, rifampin, ethambutol and pyrazinamide 19% developed ALT elevations above 10 times ULN, several polymorphisms of PXR [but not CAR] were associated with a lower incidence of liver injury).
- Dheda K, Gumbo T, Maartens G, Dooley KE, Murray M, Furin J, Nardell EA, Warren RM. Lancet Respiratory Medicine drug-resistant tuberculosis Commission group. The Lancet Respiratory Medicine Commission: 2019 update: epidemiology, pathogenesis, transmission, diagnosis, and management of multidrug-resistant and incurable tuberculosis. Lancet Respir Med. 2019;7:820–6. [PubMed: 31486393](Review of the epidemiology, pathogenesis, diagnosis and management of multidrug resistant tuberculosis; does not discuss hepatotoxicity of therapeutic drug regimens).
- Oscanoa T, Moscol S, Amado J. Clinical characteristics of pyrazinamide-associated hepatotoxicity in patients at a hospital in Lima, Peru. Rev Peru Med Exp Salud Publica. 2020;37:516–20. [PubMed: 33295555](Among 10 patients with suspected pyrazinamide hepatotoxicity, the average age was 41 years and average time to onset 41 days [range 11 to 130 days], 20% with rash and 40% with jaundice, hepatocellular enzyme elevations in 5, resolving in all patients).
- Zhao H, Wang Y, Zhang T, Wang Q, Xie W. Drug-induced liver injury from anti-tuberculosis treatment: a retrospective cohort study. Med Sci Monit. 2020;26:e920350. [PMC free article: PMC7077058] [PubMed: 32145061](Among 140 patients with liver injury due to antituberculosis therapy seen between 2009 and 2015, the median age was 40, time to onset ranged from 7-90 days [mean=24], 35% had fever, 16% rash, 68% were hepatocellular, 21% were severe and 7% died; most patients had received isoniazid, rifampin and pyrazinamide).
- Soni H, Kumar-M P, Mishra S, Bellam BL, Singh H, Mandavdhare HS, Medhi B, et al. Risk of hepatitis with various reintroduction regimens of anti-tubercular therapy: a systematic review and network meta-analysis. Expert Rev Anti Infect Ther. 2020;18:171–9. [PubMed: 31923369](Systematic review of the risk of recurrence of hepatotoxicity on reintroduction of antituberculosis agents after occurrence of drug induced liver injury indicated that sequential and incremental introduction is preferable to rechallenge with both agents concurrently).
- CDC. https://www
.cdc.gov/tb/ (CDC website with up-to-date recommendations on therapy of tuberculosis).
- PMCPubMed Central citations
- PubChem SubstanceRelated PubChem Substances
- PubMedLinks to PubMed
- Inactive hepatitis B surface antigen carrier state and hepatotoxicity during antituberculosis chemotherapy.[Chest. 2005]Inactive hepatitis B surface antigen carrier state and hepatotoxicity during antituberculosis chemotherapy.Lee BH, Koh WJ, Choi MS, Suh GY, Chung MP, Kim H, Kwon OJ. Chest. 2005 Apr; 127(4):1304-11.
- Review Ethambutol.[LiverTox: Clinical and Researc...]Review Ethambutol.. LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. 2012
- National survey to measure rates of liver injury, hospitalization, and death associated with rifampin and pyrazinamide for latent tuberculosis infection.[Clin Infect Dis. 2005]National survey to measure rates of liver injury, hospitalization, and death associated with rifampin and pyrazinamide for latent tuberculosis infection.McElroy PD, Ijaz K, Lambert LA, Jereb JA, Iademarco MF, Castro KG, Navin TR. Clin Infect Dis. 2005 Oct 15; 41(8):1125-33. Epub 2005 Sep 9.
- Risk factors for side-effects of isoniazid, rifampin and pyrazinamide in patients hospitalized for pulmonary tuberculosis.[Eur Respir J. 1996]Risk factors for side-effects of isoniazid, rifampin and pyrazinamide in patients hospitalized for pulmonary tuberculosis.Schaberg T, Rebhan K, Lode H. Eur Respir J. 1996 Oct; 9(10):2026-30.
- Review The hepatic toxicity of antituberculosis regimens containing isoniazid, rifampicin and pyrazinamide.[Tubercle. 1978]Review The hepatic toxicity of antituberculosis regimens containing isoniazid, rifampicin and pyrazinamide.Girling DJ. Tubercle. 1978 Mar; 59(1):13-32.
- Pyrazinamide - LiverToxPyrazinamide - LiverTox
Your browsing activity is empty.
Activity recording is turned off.
See more...